 |
 |
| Korean J Intern Med > Volume 11(1); 1996 > Article |
|
Abstract
Objectives :
This study was conducted to investigate the clinical features of ulcerative colitis in Korea and to evaluate the clinical course after medical therapy.
Methods :
Symptoms, signs and results of the treatment were retrospectively analyzed in 66 patients (male 32, female 34) diagnosed to have ulcerative colitis at the Asan Medical Center.
Results :
The median age of the beginning of symptoms was 36 years (range, 14ŌĆō72). Diarrhea and rectal bleeding were observed in 95.1 and 91.4%, respectively, at the time of diagnosis, while extra-colonic manifestations were observed in 24.1%. In 41 patients (62.1%), colitis developed in the rectum and sigmoid colon, while left colitis and extensive colitis developed in 11 (16.7%) and 14 patients (21.2%), respectively. The severity of disease was determined according to the clinical criteria, resulting in 22 (33.3%) mild, 21 (31.8%) moderate and 23 (34.8%) severe diseases. The seventy was also classified as 1, 2 and 3 by sigmoido-colonoscopic findings: 1;17 patients(25.8%), 2;27(40.9%) and 22(33.3%). Among 23 patients with severe disease, 5 patients (7.6%) received total colectomy due to toxic megacolon, intractability to medical therapy, ileocolic fistula and intestinal stenosis. The severity determined by colonoscopic findings was well correlated with that determined clinically and was closely related to the severity of symptoms, levels of albumin, hemoglobin and the count of leukocyte. The median duration of symptoms before treatment was 4 weeks (range. 11ŌĆō300). All patients were treated with sulfasalazine and prednisolone. All patients with medical therapy, except 2 patients (96. 7%), obtained clinical remission. The median days required for remission was 14 (range, 3ŌĆō70). Relapse rates at 6 months, 1 year and 2 years after the initiation of treatment were 19.7, 34.1 and 49.3%, respectively. The median disease-free interval from the time of remission was 10 months (range, 2ŌĆō60). After remission, the subsequent relapse rate increased in severe disease, while no difference was observed between the disease extents.
Ulcerative colitis is an inflammatory disorder of the colon, extending from the rectum, proximal to the variable length of the colon, with a chronic course of remission and relapse. Although ulcerative colitis is known to be a worldwide disorder, the prevalence of ulcerative colitis is different between races. Ulcerative colitis has primarily been a disease of whites, such as Europians and Americans. The incidence of ulcerative colitis has been very low in Korea. Over the past two decades. however, ulcerative colitis has been increasing in Korea, although the reason is still unclear1).
On the other hand, despite the recent advances in therapeutic results, medical intractability, complications and frequent relapse still remain the serious problem once ulcerative colitis develops. Because of the recent increase in the development of ulcerative colitis in Korea, it is of great importance to evaluate its clinical characteristics. The aims of the present study are to analyze clinical manifestations of ulcerative colitis, to evaluate the response to medical therapy and to investigate the factors influencing the remission and relapse in patients treated in our hospital.
Sixty-six patients with ulcerative colitis treated at the Asan Medical Center, University of Ulsan for more than 6 months from June 1989 to April 1995, were retrospectively reviewed. Thirty-two patients were males and 34 were females with the median age of 40 years at the first visit to our hospital(range 14ŌĆō72). Diagnosis of ulcerative colitis was made by characteristic clinical manifestations and endoscopic and radiologic findings. The criteria of diagnosis included the presence of gastrointestinal symptoms, such as chronic mucoid and/or bloody stool and abdominal pain, typical findings of barium enema, sigmoido-colonoscopic findings, biopsy specimens compatible with ulcerative colitis and exclusion of other colonic diseases, such as CrohnŌĆÖs disease, intestinal tuberculosis and BehcetŌĆÖs colitis, etc.
We reviewed and analyzed data on patients with ulcerative colitis according to the symptoms, signs and laboratory data of hemoglobin level, leukocyte and platelet counts in the peripheral blood, erythrocyte sedimentation rate (ESR) and serum levels of transaminases, protein and albumin. In all patients, the severity of the disease was determined by both clinical manifestations and endoscopic evidence of active inflammations. The patients were divided into three groups, mild, moderate and severe, according to the criteria proposed by Truelove and Witts with some modifications2). The clinical criteria for determining the severity at attack was as follows : mild : diarrhea<4/day with or without blood and pus in stool and no systemic symptoms, moderate : intermediate between mild and severe and severe : diarrhea>6/day, daily stool blood or systemic symptoms, such as fever, Ōēź37.5┬░C, tachycardia, Ōēź90/min, anemia and increased ESR. The severity was also graded by colonoscopic findings and scored as 1, 2 and 3 according to the cirteria reported by Gomes et al.3): 1 : mild inflammation with loss of vascular pattern, granularity or localized aphthous ulcers, 2 : severe inflammation with contact bleeding and 3 : very severe disease with friability, ulcers or spontaneous bleeding.
All patients with ulcerative colitis were divided by the extent of inflammation into four groups : proctitis, distal colitis, left colitis and extensive colitis and were diagnosed when the inflammation was localized in the rectum, from rectum to sigmoid junction, from rectum to splenic flexure and from rectum beyond splenic flexure, respectively.
The treatment results were assessed by the improvement of clinical manifestations and divided into two groups : active disease and disease in remission. Remission was defined as bowel movements Ōēż2/day and no symptoms and signs of ulcerative colitis. The relapse was defined as diarrhea Ōēź 5/day for 3 days or more. The time required for remission and disease free interval were defined as the duration from the attack to the remission and from the remission to the next relapse, respectively. The relapse rate (%) was determined 6 months, 1 year and 2 years after the remission.
Statistical significance of the difference was determined using the computer program of Spearman Correlation Coefficient, Cox regression, linear multiple regression and Kaplan-Meyer and Kruskall-Wallis 1-way ANOVA. Differences were considered to be significant when the p value was <0.05.
Clinical features of 66 patients, diagnosed to have ulcerative colitis at the Asan Medical Center, were retrospectively analyzed. The median age at the first attack was 36 years (range, 14ŌĆō72) with a slight female predominance (M : F, 1:1.1). Age and sex distribution are shown in Fig. 1, demonstrating that the incidence was the highest in the fourth decade and very low before the age of 10 and after 60 years. No previous history of ulcerative colitis was found in 36 patients, while 30 patients had the relapsed disease. The median duration of symptoms before the diagnosis was 4 weeks (range, 1ŌĆō300). The median period of follow-up was 24 months (range, 6ŌĆō60).
The clinical symptoms and signs are summarized in Table 1. Intestinal symptoms and signs were observed in all patients with the two most common symptoms being diarrhea and stool blood. In 16 patients (24.2%), extra-instestinal manifestations were observed with arthralgia being the most frequent symptom. Nine (39.1%) of 23 patients with severe disease had extra-intestinal manifestations- while none (0%) of 22 mild and 6 (28.6%) of 21 moderate disease had extraintestinal manifestations (p<0.05). The extraintestinal manifestations were not different between the disease extents.
Proctitis and left colitis were relatively common and observed in 41 (62.1%) out of 66 patients (Fig. 2). Among 66 patients, 64 had proctitis by colonoscopic and histologic findings, whereas the rectum was clearly uninvolved in 2 patients. The severity determined by colonoscopic findings was analyzed and severe disease was found more frequently with the progression of the extent (r = 0.54, p<0.01) (Table 2). The severity of disease was evaluated in all patients by both clinical symptoms and endoscopic findings. The severity, clinically determined, was well correlated to that by colonoscopic appearance (Table 3) (r = 0.91, p<0.01). In an attempt to find factors associated with the disease severity, various laboratory parameters, mentioned in the Materials and Methods, were analyzed, demonstrating that serum level of albumin and hemoglobin and leukocyte count in the peripheral blood were observed to be associated with the severity (p<0.05).
After the diagnosis, all patients were treated with sulfasalazine, while prednisolone was combined in 24 patients (36.3%). The dose of prednisolone was 40ŌĆō60 mg/day for initial attack or relapse and 5ŌĆō10 mg/day for maintenance if necessary. Patients with severe disease were treated with prednisolone more frequently (p<0.05) (Fig. 3). Five patients received operations during the medical therapy due to toxic megacolon, intractability to medical therapy, ileocolic fistula and intestinal obstruction in 1, 2, 1 and 1 patients, respectively.
Among 61 patients treated with medical therapy alone, 59 patients (96.7%) achieved remission. The median period required for the remission was 14 days (range, 3ŌĆō70). In 2 cases, symptoms were improved by the medical therapy but did not reach to the remission criteria. The median disease free interval after the remission was 10 months (range, 2ŌĆō60). The relapse increased with time and was 19.7, 34.1 and 49.3 in 6 months, 1 year and 2 years after remission. The relapse rates increased with the increase in the disease severity (p<0.05), (Fig. 4). On the other hand, no significant difference in the relapse rates was observed between the disease extents (p>0.05) (Fig. 5).
Patients with severe ulcerative colitis showed a longer period to achieve remission and a shorter disease-free interval, compared with those with the mild disease (p<0.05) (Table 4). The time to reach remission and the disease free interval were not significantly different between the disease extents. However, when patients with proctitis and distal colitis were compared with those with left and extensive colitis, the former required a shorter period to reach remission and the longer disease-free inverval compared with those with the letter (p<0.05).
Ulcerative colitis is a systemic disease associated with a number of troublesome events. Ulcerative colitis affected whites primarily in the past, although the etiology and pathogenesis are not clearly defined at present4ŌĆō6). However, considerable evidence has been accumulated that alteration of immunity is strongly associated with the clinical implicated courses of the disease7,8).
The mean annual incidence of ulcerative colitis was known for 0.9ŌĆō15.0 per 105 in western countries9ŌĆō11). In Korea, the incidence still remains very low, although it has been increasing in recent years1). The disease extent observed in this study is similar to that of the western countries9ŌĆō13). The extensive colitis observed was 21.2% in this study, while those in western countries were reported to be from 11.0% to over 37%9ŌĆō13). The proportion of proctitis and distal colitis was 62.1% in this study, which was higher than 22.2ŌĆō54.8% reported by several other investigators in Korea1,14ŌĆō18). The disease severity was reported to be somewhat different between countries. The mild disease was 33.3% in our results, while 17.8ŌĆō68.0 and 14.5ŌĆō41.0% were published to have mild disease in western countries and in Korea,1,9ŌĆō11,14ŌĆō18), respectively.
Ulcerative colitis usually develops as a proctitis and extends proximally. In patients entered into this study, 2 patients (3.3%) did not show any inflammation in the rectum. The extension rate to the more extensive disease at 5 and 10 years was reported to be 12% and 30%, respectively, in Scotland9). The follow-up period in this study, however, was too short to make a conclusive result on this point. From these clinical aspects, it may be desirable that ulcerative colitis is regarded as a disease of various phenotypes and biological behaviors19).
The severity of the disease, determined by clinical manifestations, was well correlated with that assessed by colonoscopic findings of inflammation. In order to find the indicators representing the disease severity, a number of parameters have been investigated. Seo et al.20) have reported that disease severity is well correlated with ESR. hemoglobin and serum albumin. However, Gomes et al.3) have failed to find the parameters to be correlated with the severity, although they have analyzed numerous parameters, including C-reactive protein (CRP), ESR, leucocyte and platelet counts and serum albumin. In this study, we found that serum albumin, hemoglobin and leucocyte counts were significant correlated to the severity.
The incidence of extracolonic diagnosis was reported to be 21ŌĆō45% and to be related to the disease extent21,22). However, our results showed that extra-colonic manifestations were associated with the disease severity, not with the disease extent. As immune alteration has been strongly suggested as a mechanism of ulcerative colitis, it is an interesting finding that, among 16 patients with extra-colonic manifestations, 5 patients had the disease clearly related to autoimmunity.
Most patients did not require surgical treatment. The initial response of ulcerative colitis to the medical therapy was dramatic in most patients. The long-term prognosis, however. is not always good, mainly due to the high rate of relapse. Therefore maintenance therapy is needed to suppress the relapse. The remission rate, by the medical therapy in this study, was higher than those of 75.8 and 86% reported by Chang et al.1) and Sinclair et al.9), respectively.
On the other hand, the remission rate was reported to be low in patients with relapsed disease. In the Copenhagen study, one-year probability of remission by medical therapy was reported to be 70ŌĆō90%, while it was 10ŌĆō30% in patients with relapsed disease in the previous year25). The median disease-free interval in this study was 10 months, which was longer than 17 weeks reported by Riley et al.23). The cumulative relapse rates in 2 years after remission was 49.3% in this study, while those published by other investigators were 40ŌĆō62.4%, respectively1,9).
Much attempt has been paid to investigate the factors attributed to relapse. Goodman et al24) reported that the severity of disease may be an important predictor of the relapse. To find the factors related to the relapse, we have also analysed a number of factors, such as age, sex, disease severity, disease extent, time required for remission, previous history of the disease, complete blood count, biochemical tests and acute phase reactants. We have failed, however, to find factors for predicting or reflecting the relapse.
The risk of colorectal cancer in patients with long-standing ulcerative colitis has been well documented26). To reduce the morbidity and mortality, therefore, considerable attention should be given to not only the prevention of relapse and acute complication but also to late complication such as colon cancer.
In this study, we have retrospectively analyzed the clinical characteristics of ulcerative colitis. The results have demonstrated that clinical manifestations and response to medical therapy are similar to those of western countries. To draw a conclusive result, however, the prospective study should be conducted with special emphasis on the response to therapy, relapse rate, quality of life and development of colon cancer.
Fig.┬Ā1.
Age and sex distribution of 66 patients, 32 male and 34 female, with ulcerative colitis : male : Ō¢¬ female : Ō¢Ī, total :
 .
.
 .
.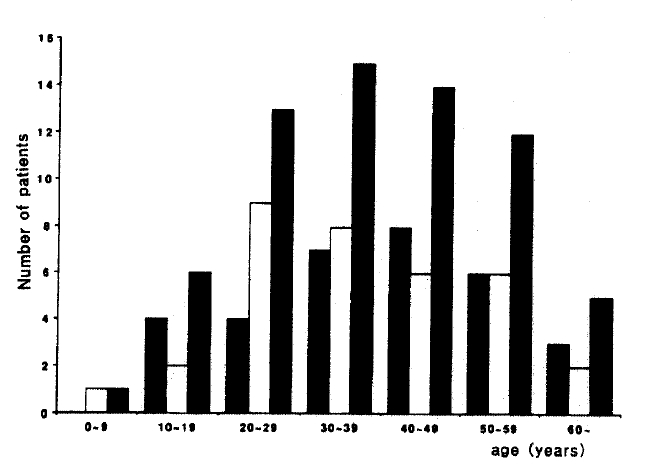
Fig.┬Ā2.
Incidence of ulcerative colitis classified by the disease extent at diagnosis : proctitis, distal colitis, left colitis and extensive colitis. * : No, of patients (%)
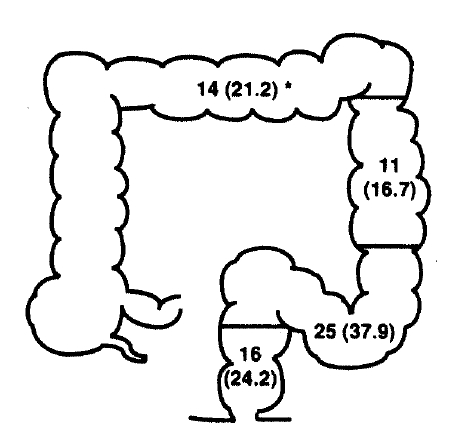
Fig.┬Ā4.
Relapse rate in 59 patients with clinically remission in ulcerative colitis medically treated according to the severity : mild :(ŌŚÅ), moderate : (*), severe : (Ō¢┤) and total : (ŌŚŗ).
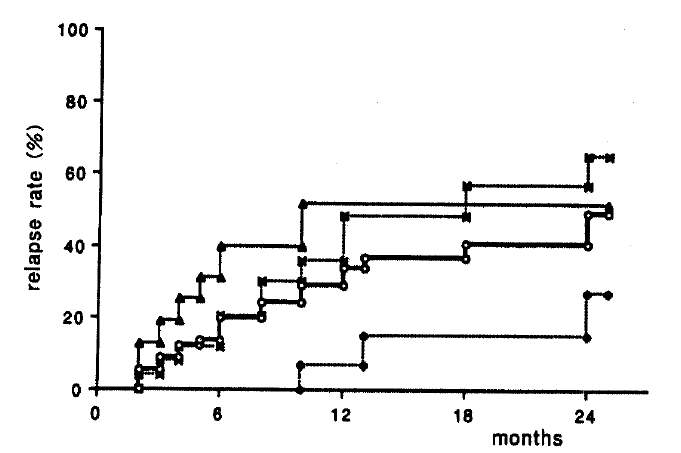
Fig.┬Ā5.
Relapse rate in 59 patients with clinically remission in ulcerative colitis according to the disease extent : proctitis (*), distal colitis (ŌŚÅ), left colitis (Ō¢Ā), extensive colitis (ŌĆó) and total (ŌŚŗ).

Table┬Ā1.
Clinical Manifestations of 66 Patients with Ulcerative Colitis
| Intestinal Manifestations | |
|---|---|
| Diarrhea | 63* (95.1) |
| Stool blood | 60 (91.4) |
| Abdominal pain | 35 (53.4) |
| Abdominal tenderness | 33 (50.0) |
| Mucus discharge | 27 (40.9) |
| Systemic symptoms | 24 (36.4) |
| Weight loss | 22 (33.3) |
| General weakness | 19 (28.7) |
| Tachycardia | 9 (13.6) |
| Fever Ōēź37.5┬░C | 9 (13.6) |
|
|
|
| Extra-intestinal manifestations | |
|
|
|
| Arthralgia | 7 (10.6) |
| Ureter stone | 2 (3.0) |
| Immune hemolytic anemia | 1 (1.5) |
| HashimotoŌĆÖs thyroiditis | 1 (1.5) |
| Idiopathic thrombocytopenic purpura | 1 (1.5) |
| Sclerosing cholangitis | 1 (1.5) |
| Central serous chorioretinitis | 1 (1.5) |
| Anxiety disorder | 1 (1.5) |
| Myalgia | 1 (1.5) |
Table┬Ā2.
Severity of Ulcerative Colitis According to Disease Extents
Table┬Ā3.
Correlations Between Disease Severity Determined by Clinical Symptoms and That by Colonoscopic Features
| Colonoscopically determined severity |
Clinical determined severity
|
|||
|---|---|---|---|---|
| Mild | Moderate | Severe | Total | |
| 1 | 17(25.7)* | 0(0.0) | 0(0.0) | 7(25.8) |
| 2 | 5(7.6) | 20(30.3) | 2(3.0) | 27(40.9) |
| 3 | 0(0.0) | 1(1.5) | 21(31.8) | 22(33.3) |
|
|
||||
| Total | 22(33.3) | 21(31.8) | 23(34.8) | 66(100.0) |
Table┬Ā4.
Responses to Medical Therapy According to Disease Severity and Extent
| No. Patients | Time to Remission (days) | Disease Free Interval (months) | |
|---|---|---|---|
| Severity | |||
| ŌĆāŌĆāmild | 17 | 13.4┬▒1.7* | 19.8┬▒3.2 |
| ŌĆāŌĆāmoderate | 26 | 13.6┬▒1.6 | 12.6┬▒2.1 |
| ŌĆāŌĆāsevere | 16 | 32.6┬▒4.3 | 9.2┬▒2.1 |
| ŌĆāŌĆāp value | <0.01 | <0.05 | |
| Extent | |||
| ŌĆāŌĆāproctitis | 16 | 11.6┬▒1.6 | 13.4┬▒1.9 |
| ŌĆāŌĆādistal colitis | 24 | 16.8┬▒2.9 | 16.5┬▒2.9 |
| ŌĆāŌĆāleft colitis | 8 | 27.4┬▒7.1 | 10.1┬▒3.0 |
| ŌĆāŌĆāextensive colitis | 11 | 27.2┬▒5.8 | 10.5┬▒3.3 |
| ŌĆāŌĆāp value | >0.05 | >0.05 |
REFERENCES
1. Chang DK, Lee KL, Kim JG, Kim YT, June HC, Song IS, Choi KW, Kim CY, Park JG, Rhee PL, Choi SW. Follow-up of ulcerative colitis: Short-term outcome to medical treatment and relapse rates. Kor J Gastroenterol 1994;26(6):907ŌĆō918.
3. Gomes P, Boulay CD, Smith CL, Holdstock G. Relationship between disease activity indicies and colonoscopic findings in patients with colonic inflammatory bowel disease. Gut 1986;27:92ŌĆō95.



4. Gitnick G. Etiology of inflammatory bowel disease: Where have we been? Where are we doing? Scand J Gastroenterol 1990;25(Suppl 175):93ŌĆō96.

6. Shanahan F, Duerr RH, Rotter JI, Yang H, Sutherland LR, Mceiree C, Landers CJ, Targan SR. Neutrophil autoantibodies In ulcerative colitis: Familial aggression and genetic heterogeneity. Gastroenterology 1992;103:456ŌĆō461.


7. Matsuura TM, West GA, Youngman KR, Klein JS, Fiocchi C. Immune activation genes in inflammatory bowel disease. Gastroenterology 1993;104:448ŌĆō458.


8. Mayer L, Eisenhardt D. Lack of induction of suppressor T cells by intestinal epithelial cells from patients with inflammatory bowel disease. J Clin invest 1990;86:1255ŌĆō1260.



9. Sinclair TS, Brunt PW, Mowat NA. Nonspecific proctocolitis northeastern Scotland: A community study. Gastroenterology 1983;85(1):1ŌĆō11.


10. Stonnington CM, Phillips SF, Zinsmeister AR, Melton LJ. Chronic ulcerative colis: Incidence and prevalence in a community. Gut 1987;28:402ŌĆō409.



11. Langholz E, Munkholm P, Nielsen OH, Kreiner S, Binder V. Incidence and prevalence of ulcerative colitis in Copenhagen from 1962 to 1987. Scand J Gastroenterol 1991;26:1247ŌĆō1256.


12. Richie JK, Powell-Tuck J, Lennard-Jones JE. Clinical outcome of the first ten years of ulcerative colitis and proctits. Lancet 1978;1:1140ŌĆō1143.


13. Brostrom O, Monsen U, Nordenwall B, Sorstad J, Hellers G. Prognosis and mortality of ulcerative colitis in Stockholm county, 1955ŌĆō1979. Scand J Gastroenterol 1987;22:907ŌĆō913.


14. Back KH, Jun KY. A clinical review of ulcerative colitis. Kor J Surg 1988;35(6):690ŌĆō697.
16. Choi JH, Hyun JH. A clinical study on 39 cases of ulcerative colitis. Kor J Gastroenterol 1992;24(3):493ŌĆō500.
17. Yang US. Clinical observation of ulcerative colitis. Pusan Med Coll 1989;29(2):145ŌĆō152.
18. Kwon KH, Park CS, Kim BR, Kim CK. A clinical review of ulcerative colitis. Kor J Gastreonterol 1984;16(1):121ŌĆō127.
19. Jenkins D, Goodall A, Scott B. Ulcerative colitis: One disese or two? (quantitative histological differences between distal and extensive disease). Gut 1990;31:426ŌĆō430.



20. Seo M, Okada M, Yao T, Ueki M, Arima S, Okumura M. An index of disease activity in patients with ulcerative colitis. Am J Gastroenterol 1992;87(8):971ŌĆō976.

21. Monsen U, Sorstad J, Hellers G, Johansson C. Extracolonic diagnoses in ulcerative colitis: An epidemiological study. Am J Gastroenteral 1990;85(6):711ŌĆō716.
22. Greenstein AJ, Janowitz HD, Sachar DB. The extra-intestinal complications of CrohnŌĆÖs disease and ulcerative colitis: A study of 700 patients. Medicine 1976;55(4):401ŌĆō412.


23. Riley SA, Mani V, Goodman MJ, Lucas S. Why do patients with ulcerative colitis relapse? Gut 1990;31:179ŌĆō183.



24. Goodmann MW, Eberle DE, Kuchler LA, Fossum EA. Microsomal activity predicts in patients with distal ulcerative colitis on 4-aminosalicylic acid enemas (abstract). Gastroenterology 1988;94:A151.

-
METRICS

- Related articles
-
Current state and prospects of gout treatment in Korea2022 July;37(4)
Clinical course of severe esophagitis following bevacizumab therapy2022 January;37(1)
Current status of pulmonary arterial hypertension in Korea2019 July;34(4)
Clinical features of snoring patients during sedative endoscopy2019 March;34(2)




 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print


