1. Hakim R, Himmelfarb J. Hemodialysis access failure: a call to action. Kidney Int 1998;54:1029ŌĆō1040.


2. Stracke S, Konner K, Kostlin I, et al. Increased expression of TGF-beta1 and IGF-I in inflammatory stenotic lesions of hemodialysis fistulas. Kidney Int 2002;61:1011ŌĆō1019.


3. Sterpetti AV, Cucina A, Santoro L, Cardillo B, Cavallaro A. Modulation of arterial smooth muscle cell growth by haemodynamic forces. Eur J Vasc Surg 1992;6:16ŌĆō20.


4. Himmelfarb J. Pharmacologic prevention of vascular access stenosis. Curr Opin Nephrol Hypertens 1999;8:569ŌĆō572.


5. Sreedhara R, Himmelfarb J, Lazarus JM, Hakim RM. Anti-platelet therapy in graft thrombosis: results of a prospective, randomized, double-blind study. Kidney Int 1994;45:1477ŌĆō1483.


6. Kaufman JS, O'Connor TZ, Zhang JH, et al. Randomized controlled trial of clopidogrel plus aspirin to prevent hemodialysis access graft thrombosis. J Am Soc Nephrol 2003;14:2313ŌĆō2321.


7. Saran R, Dykstra DM, Wolfe RA, et al. Association between vascular access failure and the use of specific drugs: the Dialysis Outcomes and Practice Patterns Study (DOPPS). Am J Kidney Dis 2002;40:1255ŌĆō1263.


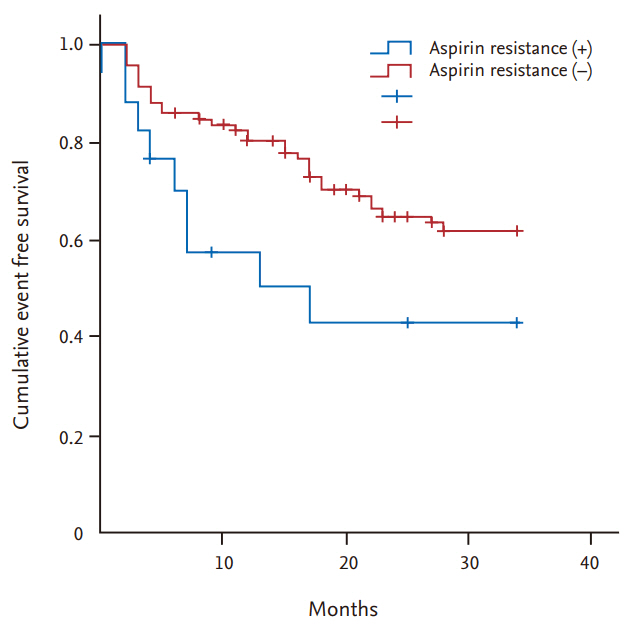
8. Kilickesmez KO, Kocas C, Abaci O, Okcun B, Gorcin B, Gurmen T. Follow-up of aspirin-resistant patients with end-stage kidney disease. Int Urol Nephrol 2013;45:1097ŌĆō1102.


9. Bath PM, Missouris CG, Buckenham T, MacGregor GA. Increased platelet volume and platelet mass in patients with atherosclerotic renal artery stenosis. Clin Sci (Lond) 1994;87:253ŌĆō257.


10. Tschope D, Langer E, Schauseil S, Rosen P, Kaufmann L, Gries FA. Increased platelet volume: sign of impaired thrombopoiesis in diabetes mellitus. Klin Wochenschr 1989;67:253ŌĆō259.


11. Valkila EH, Salenius JP, Koivula TA. Platelet indices in patients with occlusive carotid artery disease. Angiology 1994;45:361ŌĆō365.


12. D'Erasmo E, Aliberti G, Celi FS, Romagnoli E, Vecci E, Mazzuoli GF. Platelet count, mean platelet volume and their relation to prognosis in cerebral infarction. J Intern Med 1990;227:11ŌĆō14.


13. Martin JF, Bath PM, Burr ML. Influence of platelet size on outcome after myocardial infarction. Lancet 1991;338:1409ŌĆō1411.


14. Ibels LS, Stewart JH, Mahony JF, Neale FC, Sheil AG. Occlusive arterial disease in uraemic and haemodialysis patients and renal transplant recipients: a study of the incidence of arterial disease and of the prevalence of risk factors implicated in the pathogenesis of arteriosclerosis. Q J Med 1977;46:197ŌĆō214.

15. Lindner A, Charra B, Sherrard DJ, Scribner BH. Accelerated atherosclerosis in prolonged maintenance hemodialysis. N Engl J Med 1974;290:697ŌĆō701.


17. Jakubowski JA, Thompson CB, Vaillancourt R, Valeri CR, Deykin D. Arachidonic acid metabolism by platelets of differing size. Br J Haematol 1983;53:503ŌĆō511.


18. Martin JF, Trowbridge EA, Salmon G, Plumb J. The biological significance of platelet volume: its relationship to bleeding time, platelet thromboxane B2 production and megakaryocyte nuclear DNA concentration. Thromb Res 1983;32:443ŌĆō460.


22. Irish AB, Viecelli AK, Hawley CM, et al. Effect of fish oil supplementation and aspirin use on arteriovenous fistula failure in patients requiring hemodialysis: a randomized clinical trial. JAMA Intern Med 2017;177:184ŌĆō193.


23. Murley A, Wijewardane A, Wilmink T, Baharani J. Should patients be on antithrombotic medication for their first arteriovenous fistulae? J Vasc Access 2016;17:118ŌĆō123.


24. Guirgis M, Thompson P, Jansen S. Review of aspirin and clopidogrel resistance in peripheral arterial disease. J Vasc Surg 2017;66:1576ŌĆō1586.


26. Liu L, Gao YH, Cao J, et al. High prevalence of aspirin resistance in elderly patients with cardiovascular disease and metabolic syndrome. J Geriatr Cardiol 2016;13:531ŌĆō536.


27. Li J, Song M, Jian Z, et al. Laboratory aspirin resistance and the risk of major adverse cardiovascular events in patients with coronary heart disease on confirmed aspirin adherence. J Atheroscler Thromb 2014;21:239ŌĆō247.


28. Kim HW, Jang EH, Kim SY, Kim KS, Joo SJ. Aspirin resistance in patients with end-stage renal disease. Korean J Nephrol 2011;30:163ŌĆō170.
29. Sansanayudh N, Muntham D, Yamwong S, Sritara P, Akrawichien T, Thakkinstian A. The association between mean platelet volume and cardiovascular risk factors. Eur J Intern Med 2016;30:37ŌĆō42.


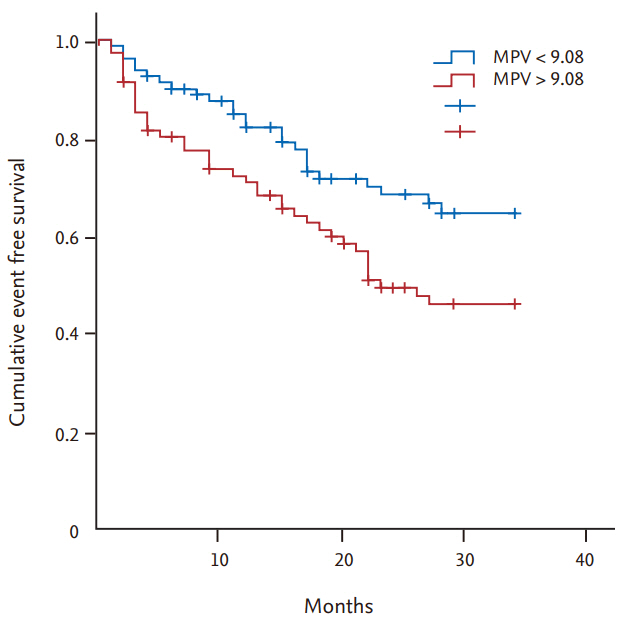
30. Varol E, Ozaydin M. Mean platelet volume measurement in chronic renal failure: confounding factors must have been taken into account. Ren Fail 2014;36:488.


31. Henning BF, Zidek W, Linder B, Tepel M. Mean platelet volume and coronary heart disease in hemodialysis patients. Kidney Blood Press Res 2002;25:103ŌĆō108.


32. Colkesen Y, Coskun I, Muderrisoglu H. The effect of aspirin on mean platelet volume in patients with paroxysmal atrial fibrillation. Platelets 2013;24:263ŌĆō266.


33. Vinholt PJ, Hvas AM, Frederiksen H, Bathum L, Jorgensen MK, Nybo M. Platelet count is associated with cardiovascular disease, cancer and mortality: a population-based cohort study. Thromb Res 2016;148:136ŌĆō142.


34. Levin J, Bessman JD. The inverse relation between platelet volume and platelet number. Abnormalities in hematologic disease and evidence that platelet size does not correlate with platelet age. J Lab Clin Med 1983;101:295ŌĆō307.

35. Gheith OA, Kamal MM. Risk factors of vascular access failure in patients on hemodialysis. Iran J Kidney Dis 2008;2:201ŌĆō207.

37. Yazici M, Kaya A, Kaya Y, Albayrak S, Cinemre H, Ozhan H. Lifestyle modification decreases the mean platelet volume in prehypertensive patients. Platelets 2009;20:58ŌĆō63.


40. Azab B, Torbey E, Singh J, et al. Mean platelet volume/platelet count ratio as a predictor of long-term mortality after non-ST-elevation myocardial infarction. Platelets 2011;22:557ŌĆō566.








 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print


