INTRODUCTION
Rheumatoid arthritis (RA) is a chronic autoimmune disease characterized by persistent inflammation of the joint synovium and synovial hyperplasia. The aberrant changes, which occur in the synovium, lead to joint damage and functional disability. Although the pathogenesis of RA has not been clearly identified, it is clear that a cascading network of pro-inflammatory cytokines plays a major role in the development of RA [1]. Toll-like receptor 4 (TLR4) contributes to the induction of pro-inflammatory cytokines by activating innate immunity under certain circumstances [2]. To date, several studies have suggested that TLR4 is implicated in the pathogenesis of RA. TLR4 is highly expressed in the inflamed synovium of RA patients [3]. The activation of the TLR4 signaling pathway is involved in persistent inflammation and joint destruction in RA [4]. Associations between TLR4 single nucleotide polymorphisms and RA disease activity have been reported [5].
Grape seed proanthocyanidin extract (GSPE) is a standardized water-ethanol extract derived from red grape seeds in which various antioxidants including catechins and oligomeric proanthocyanidins are accumulated [6]. GSPE is made up of a combination of ingredients with 15% (+) catechin and (–) epicatechin; 80% (–) epicatechin 3-O-gallate, dimers, trimers, tetramers, and their gallates; and 5% pentamers, hexamers, heptamers, and their gallates [7]. Proanthocyanidins are the most abundant phenolic compounds in grape seeds. They have been reported to have various bio-protective functions such as anti-bacterial, anti-viral, anti-allergic, anti-carcinogenic, anti-thrombotic, and vasodilatory activities [8]. Proanthocyanidins possess more potent anti-oxidative activity than vitamins C, E, and β-carotene [9]. Furthermore, the anti-inflammatory properties of proanthocyanidins on experimental inflammation, including in animal models of ulcerative colitis, skin tumors, and collagen-induced arthritis (CIA), have been shown in previous studies [10-13]. However, little information is available concerning how proanthocyanidins regulate experimental autoimmune arthritis. In this study, we hypothesized that GSPE would influence TLR4 signaling pathways, which play a critical role in the pathogenesis of RA, in the development of murine autoimmune arthritis. We also examined the immunological effect of GSPE on fibroblast-like synoviocytes (FLSs) in patients with RA.
METHODS
Experimental animals
Dilute brown non-agouti (DBA)/1J mice (6 to 8 weeks old; SLC Inc., Hamamatsu, Japan), were housed in polycarbonate cages and fed with standard mouse chow (Ralston Purina, St. Louis, MO, USA) and water ad libitum. All experimental procedures were examined and approved by the Animal Research Ethics Committee of the Keimyung University (No. KM-2009-06R).
Induction of CIA and GSPE treatment
Male DBA/1J mice were given an intradermal injection of 100 μg of bovine type II collagen emulsified in complete Freund’s adjuvant (1:1, w/v; Chondrex, Redmond, WA, USA) into the base of the tail. Three weeks after primary immunization, a booster, which consisted of 100 μg of bovine type II collagen emulsified in incomplete Freund’s adjuvant (IFA; 1:1, v/v; Chondrex), was injected intradermally. Mice were divided into three groups; control, CIA, and CIA with GSPE treatment. To investigate the effects of GSPE on the development of arthritis, CIA mice were treated with GSPE (100 mg/kg body weight) five times per week for 3 weeks intraperitoneally, after the booster injection. GSPE was kindly provided by Hanlim Pharmaceutical Company (Seoul, Korea). GSPE dissolved in saline was used for the experiments.
Clinical arthritis severity score
Three observers scored the severity of arthritis independently from day 24 after primary immunization to day 42. Clinical arthritis scores of the mice were evaluated as follows: 0, no evidence of swelling; 1, mild swelling confined to the ankle or the midfoot (tarsals); 2, moderate swelling extending from the ankle to the midfoot (tarsals); and 3, severe swelling and erythema extending from the ankle to the metatarsal joints or ankylosis of the ankle joint. The sum of the scores of four limbs represented the arthritis score of each mouse.
Histological analysis
Joint tissues were fixed with 4% paraformaldehyde. They were then decalcified for 1 week in hydrochloric acid before being embedded in paraffin. Tissue sections (4 μm) were stained with H&E in order to determine the influx of inflammatory cells. Inflammation was scored using the following criteria: 0, no inflammation; 1, slight thickening of lining layer or some infiltrating cells in sublining layer; 2, slight thickening of lining layer plus some infiltrating cells in sublining layer; 3, thickening of lining layer, influx of cells in sublining layer and presence of cells in the synovial space and synovium highly infiltrated with many inflammatory cells. The tissue sections were stained with safranin O to evaluate cartilage degradation under light microscopic examination. The extent of cartilage damage scored using the following criteria: 0, no destruction; 1, minimal erosion limited to single spots; 2, slight to moderate erosion in a limited area; 3, more extensive erosion and destruction. Immunohistochemistry was performed using the Vectastain ABC kit and 3,3’-diaminobenzidine as a substrate (both from Vector Laboratories, Burlingame, CA, USA). Nonspecific staining was reduced by protein blocking with 20% normal goat serum for 30 minutes, at room temperature. Joint tissues were incubated with anti-TLR4 antibody (Santa Cruz Biotechnology, Dallas, TX, USA) overnight at 4ºC.
Enzyme-linked immunosorbent assay
The serum levels of anti-type II collagen specific immunoglobulin G (IgG; Chondrex), tumor necrosis factor α (TNF-α), interleukin 6 (IL-6), and IL-17 (R&D Systems, Minneapolis, MN, USA) were measured according to the manufacturer’s protocol using an enzyme-linked immunosorbent assay (ELISA) kit (R&D). For the determination of cytokine levels, blood samples from the mice were obtained on day 42 and stored at –70°C until use. Antibody concentrations for each serum sample were obtained by reference to the standard curve. The absorbance values were determined at 450 nm with an ELISA microplate reader (Vector3, Perkin Elmer, Waltham, MA, USA).
Protein extraction and Western blot analysis
Ankles joints of mice were isolated by disarticulation distal to tibia. Synovial tissues from hind paws were collected and frozen until experiments. The frozen synovial tissues were pulverized and synovial protein was extracted as follows. Pulverized synovial tissues were lysed in ice-cold lysis buffer (pH 7.4, 50 mM Tris-HCl, 1% Nonidet P-40, 0.25% sodium deoxycholate, 150 mM NaCl, 1 mM Na3VO4, and 1 mM NaF) containing protease inhibitors (2 mM phenylmethylsulfonyl fluoride, 100 μg/mL leupeptin, 10 μg/mL pepstatin, 1 μg/mL aprotinin, and 2 mM ethylenediaminetetraacetic acid). Cytoplasmic and nuclear extracts were prepared from joint tissues using the NE-PER nuclear and cytoplasmic protein extraction reagent kit (Thermo Pierce, Rockford, IL, USA). Samples in each group were subjected to 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis on resolving gels appropriately and transferred to a nitrocellulose membrane (Millipore, Billerica, MA, USA). Blots were incubated overnight at 4°C with primary antibodies for TLR4, myeloid differentiation factor 88 (MyD88), Toll/IL-1 receptor domain-containing adaptor inducing IFN-β (TRIF), IκBα, phosphorylated IκBα (p-IκBα), p65, p50 (all from Santa Cruz Biotechnology), and β-actin (Sigma-Aldrich, St. Louis, MO, USA). Anti-laminB1 (Santa Cruz Biotechnology) was used as an internal control of nucleus protein marker. The membrane was then washed with a mixture of Tris-buffered saline with Tween 20 (TBS-T) and incubated with horseradish peroxidase-conjugated secondary antibodies (Santa Cruz Biotechnology). Protein bands were detected using enhanced chemiluminescent reagents (Amersham Pharmacia Biotech, Uppsala, Sweden).
Isolation and culture of FLS
FLS were isolated by enzymatic digestion of synovial tissue, provided by the Keimyung Human Bio-Resource Bank, a member of the National Biobank of Korea. The tissue was obtained from patients with osteoarthritis (OA) and from those with RA who underwent total joint replacement surgery or a synovectomy at Keimyung University Dongsan Medical Center. All the patients fulfilled the American College of Rheumatology criteria and agreed to give their informed consent (approved by the Institutional Review Board [IRB] of Keimyung University Dongsan Medical Center [IRB No. 2015-02-005]) [14]. The tissue samples were minced into 2 to 3 mm pieces and treated for 4 hours with 0.5 mg/mL type II collagenase (Worthington Biochemical Corp., Lakewood, NJ, USA) in Dulbecco′s modified Eagle′s medium (DMEM) at 37ºC in a water bath. Synoviocytes were grown in DMEM supplemented with 10% fetal bovine serum, 2 mM L-glutamine, 100 units/mL penicillin, and 100 ng/mL streptomycin. The cells were used for experiments from passage 4 to 8, at which time the population was homogeneous.
Statistical analysis
The statistical analysis was performed using the SPSS version 22 (IBM Co., Armonk, NY, USA). Student t tests and one-way analysis of variance were carried out. The accepted level of significance was preset at p < 0.05. Each experiment was executed at least three times in duplicate. Data are presented as mean ± SD.
RESULTS
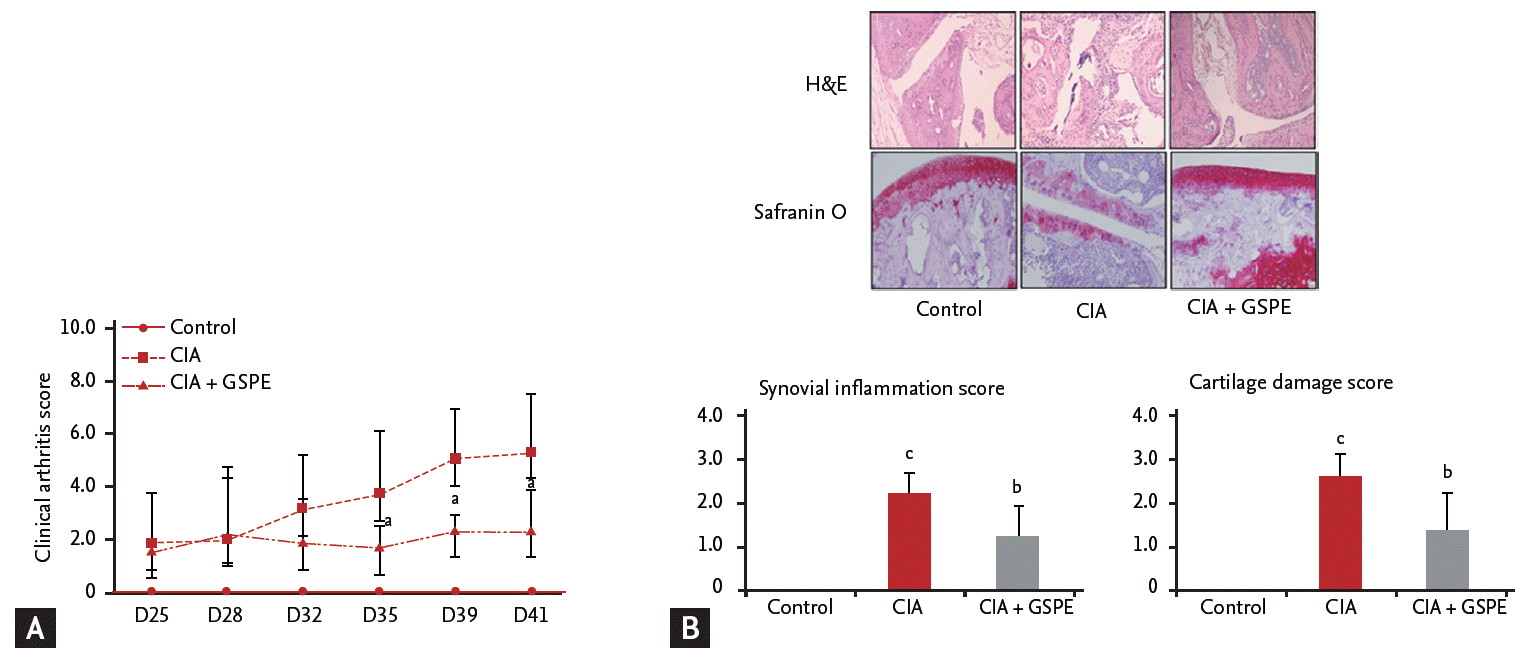
GSPE ameliorates murine autoimmune arthritis
We examined the effect of GSPE on the development of experimental arthritis in vivo using a CIA mouse model. DBA/1J mice were intraperitoneally injected with GSPE five times per week for 3 weeks after the second immunization with type II collagen/IFA. The mean arthritis score of GSPE-treated CIA mice was significantly lower than that of CIA mice (Fig. 1A). Representative histologic sections of hind paws of the mice are shown in Fig. 1B. The joints of CIA mice revealed the infiltration of inflammatory cells, synovial proliferation, and cartilage destruction (Fig. 1B). In CIA mice treated with GSPE, there was a reduction in infiltrated inflammatory cells, synovial proliferation, and cartilage damage (Fig. 1B).
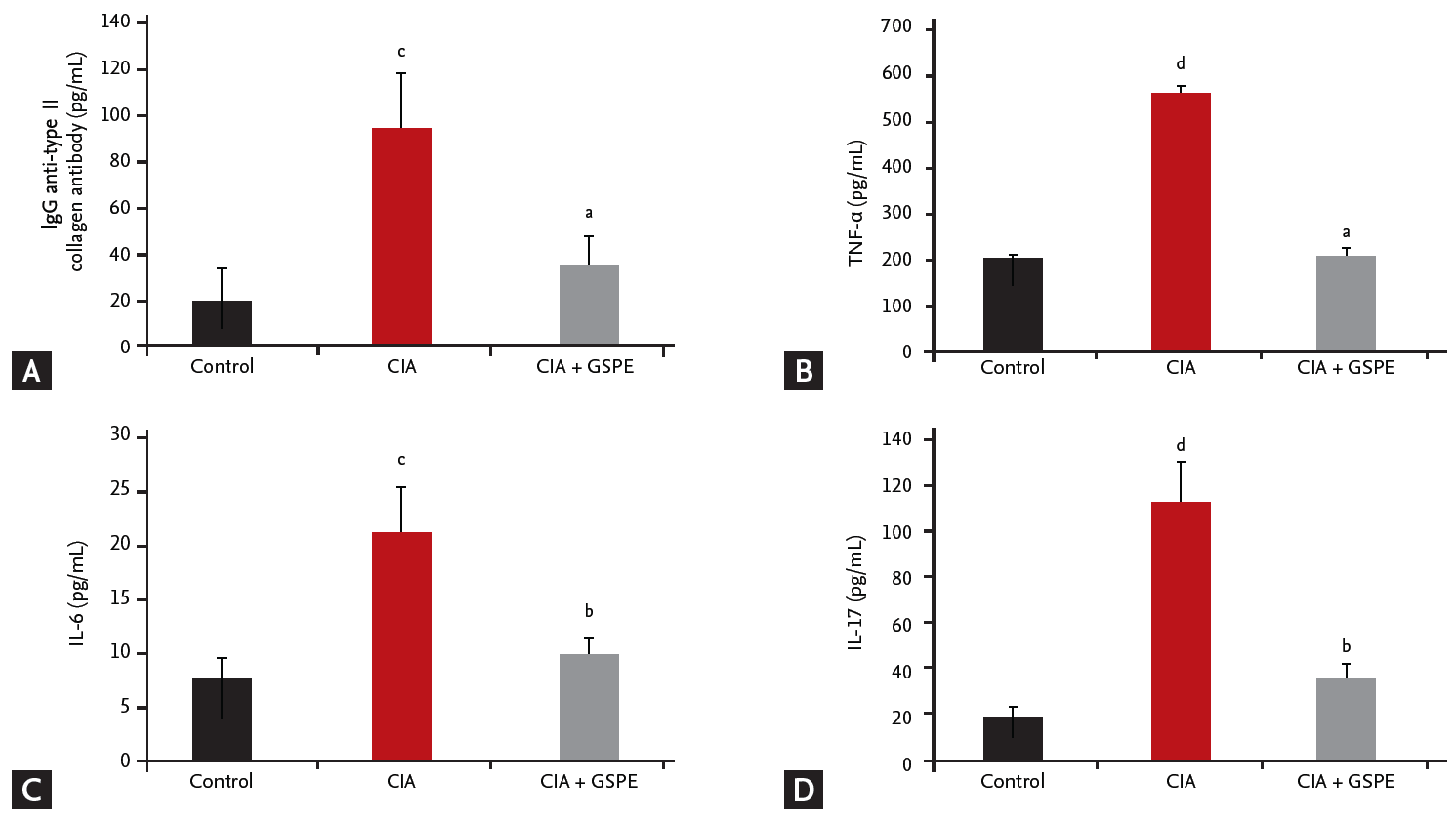
GSPE decreases serum levels of pro-inflammatory cytokines in CIA mice
We investigated whether the therapeutic effect of GSPE in CIA mice affects the humoral immune response to type II collagen and serum levels of pro-inflammatory cytokines. The levels of anti-type II collagen IgG, TNF-α, IL-6, and IL-17 in serum samples of mice were measured using ELISA, 6 weeks after primary immunization. The serum concentrations of anti-type II collagen IgG were significantly reduced in CIA mice following GSPE treatment, when compared with CIA mice (Fig. 2A). There was also a decrease in serum levels of TNF-α (Fig. 2B), IL-6 (Fig. 2C), and IL-17 (Fig. 2D) in the GSPE-treated group.
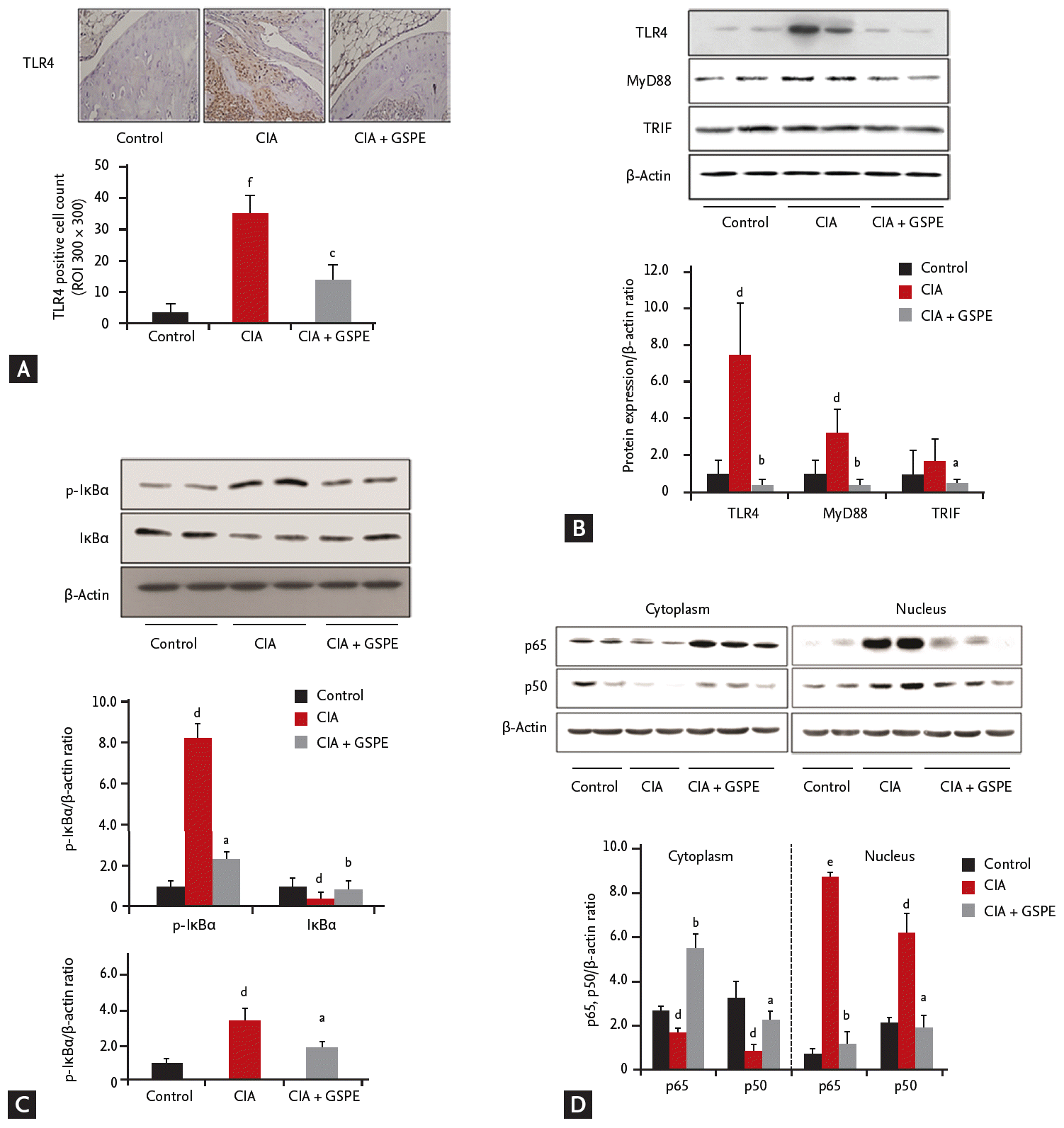
GSPE administration reduces TLR4 expression in the synovium of CIA mice
TLR4 has been reported to play a critical role in the pathogenesis of CIA [15,16]. Therefore, we investigated whether GSPE treatment influenced TLR4 expression in the synovial tissue of CIA using immunohistochemical staining. To assess the in vivo effect of GSPE on TLR4 expression, histological sections of the tibiotalar joints were stained in order to visualize the presence of TLR4. As shown in Fig. 3A, less TLR4-positive cells were observed in the joints of GSPE-treated CIA mice than in those of CIA mice. We also investigated the effect of GSPE on the intracellular proteins involved in TLR4-mediated signal transduction in the synovium of CIA, by using Western blotting. Synovial protein levels of TLR4, MyD88, and TRIF were analyzed. The expression levels of TLR4 and MyD88 proteins were upregulated in the synovial extracts of CIA mice when compared with those of control mice. TLR4 and MyD88 expression levels were downregulated in GSPE-treated CIA mice when compared with CIA mice (Fig. 3B). The expression level of TRIF showed no increase in CIA mice when compared to the control mice. However, GSPE treatment downregulated the expression level of TRIF in CIA mice when compared to CIA mice (Fig. 3B). In order to explore the effects of GSPE on the activation of nuclear factor-κB (NF-κB) signaling, the levels of IκBα and p-IκBα were measured. GSPE reduced the level of p-IκBα protein in the synovial tissue of CIA mice (Fig. 3C). The ratio of p-IκBα to total IκBα was also lower in GSPE-treated CIA mice than in CIA mice (Fig. 3C). We also examined whether GSPE affected nuclear translocation of the NF-κB subunits, p65 and p50. The nuclear extracts and cytoplasmic lysates were purified from the synovial tissue and subjected to Western blotting. The nuclear localization of p65 and p50 NF-κB was inhibited in the synovial extracts of GSPE-treated CIA mice, while nuclear localization of p65 and p50 NF-κB was prominent in those of CIA mice (Fig. 3D).
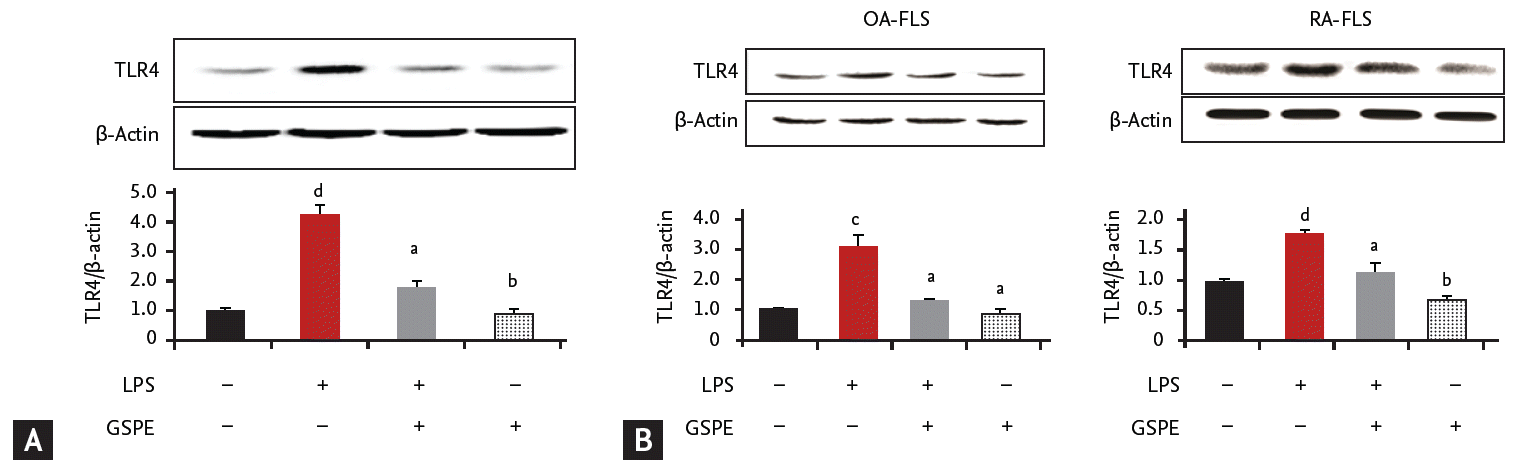
GSPE suppresses lipopolysaccharide-induced TLR4 activation in RAW264.7 cells
Our data demonstrates that GSPE administration decreased TLR4 expression in the synovium of CIA mice (Fig. 3A and 3B). We next investigated whether GSPE had an influence on TLR4 expression in vitro by using a murine macrophage cell line, RAW264.7. GSPE downregulated the expression of TLR4, which was enhanced by stimulation with lipopolysaccharide (LPS) in RAW264.7 cells (Fig. 4A). We also evaluated the effect of GSPE on the expression of TLR4 in FLS from patients with OA and RA. As shown in Fig. 4B, LPS-induced TLR4 upregulation was suppressed by GSPE in FLS from patients with RA and OA.
DISCUSSION
Proanthocyanidins, which are the main constituents of GSPE, belong to the category of condensed tannins [17]. The sources of proanthocyanidins include fruits, vegetables, nuts, seeds, flowers, and bark [18]. Proanthocyanidins are well-known as naturally occurring anti-oxidants. They have been reported to have beneficial effects on modulating inflammation in human cells, such as differentiated adipocytes and human pulmonary epithelial cells in vitro, in addition to animal inflammatory models as mentioned above [19,20]. Some previous studies have shown that GSPE has an anti-inflammatory effect in an animal model of RA. Cho et al. [13] first reported about the therapeutic effect of GSPE on CIA and showed the effects of GSPE on oxidative stress and osteoclastogenesis in vitro. Another study using a murine model of RA has demonstrated the effect of GSPE focusing on inflammation-associated bone destruction [21]. GSPE attenuated both arthritis and obesity in obese CIA mice [22]. However, little is known about the mechanism by which GSPE regulate the inflammatory response. Oxidative stress is reduced by GSPE, and has been reported to be involved in the pathogenesis of murine autoimmune arthritis. However, there has been controversy as to whether oxidative stress instigates, or suppresses the inflammation in a model of murine autoimmune arthritis [23-25]. One previous study reported that GSPE influences murine autoimmune arthritis by regulating Foxp3+ regulatory and IL-17-producing T cells, reciprocal control of which is important in managing autoimmune arthritis [26]. There was another report that GSPE has an anti-arthritic effect in adjuvant-induced arthritis model by modifying T cell subsets [27]. In this study, we focused on revealing the mechanism of action of GSPE in suppressing murine autoimmune arthritis. Here, we demonstrated that GSPE has an anti-arthritic effect through the regulation of TLR4-mediated signaling for the first time.
TLRs are innate receptors, which play an essential role in the innate immune system [28]. Among diverse TLRs, TLR4 was the first to be characterized, and has been reported to play a significant role in the pathogenesis of autoimmune diseases [29]. In particular, a large amount of research provides evidence that TLR4 is implicated in the pathogenesis of RA. TLR4-deficient mice showed lower degree of severity and incidence of CIA compared to the wild-type mice [15]. LPS-induced TLR4 activation was greatly enhanced in peripheral blood mononuclear cells (PBMCs) from patients with RA than in those from patients with OA or healthy controls [30]. TLR4 activation by LPS induces the production of pro-inflammatory cytokines in human FLS and PBMCs from patients with RA [31,32]. TLR4 upregulation increases pro-inflammatory activity and induces degeneration in chondrocytes [33]. Although controversy exists regarding the association between TLR4 polymorphisms and RA, previous studies have reported that a functional variant of TLR4 is associated with susceptibility to RA and that non-missense genetic polymorphisms of TLR4 confer the risk of the development of RA [34,35]. Our data demonstrated that TLR4 is highly expressed in the joints of CIA mice. This observation supports a previous study, which confirmed that treatment with a TLR4 antagonist decreased the severity of CIA [16]. In this study, GSPE-treated mice had fewer TLR4-expressing cells in their synovium in comparison to those that were not treated with GSPE. This suggests that GSPE inhibits TLR4 expression in the synovium in vivo. However, little is known about the effects of GSPE on the expression of TLR4 in the synovium of CIA mice. Therefore, further investigation into the mechanism by which GSPE suppresses the expression of TLR4 in the synovium is required.
TLR4 is regarded as an important receptor in inflammatory responses. TLR4 ligation leads to the activation of a downstream transcription factor, NF-κB, which is linked by MyD88, a key adaptor protein in TLR4-NF-κB signaling. The stimulation of TLR4 recruits the MyD88 adaptor protein, leading to IκB kinase activation and culminating in IκBα phosphorylation. IκBα phosphorylation releases cytosolic sequestration of p65 and p50 NF-κB subunits and causes nuclear repositioning of those proteins. In this study, we evaluated the in vivo effect of GSPE on NF-κB and MyD88. We also examined the influence of GSPE on the various steps in NF-κB activation by performing cell fractionation experiments. Our data showed that GSPE treatment diminishes the phosphorylation of IκBα and inhibits nuclear translocation of p65 and p50 NF-κB subunits. The results indicate that GSPE inhibits NF-κB activation through the suppression of IκBα phosphorylation. In addition to MyD88, TRIF is also an adaptor protein, which responds to the activation of TLR4 [36]. However, the data presented here show no significant change in the expression of TRIF between CIA and control mice. These results may imply that the inflammatory responses of CIA utilize the TLR4-MyD88-dependent pathway rather than TLR4-TRIF-dependent pathway, although further studies are required to confirm these tentative implications.
Prior to in vitro experiments detailed above, we also evaluated the cytotoxic effect of GSPE on FLS from RA or OA patients, and RAW264.7 cells using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenylphenyltetrazolium bromide (MTT) assay. After a 24-hour incubation period, GSPE, at concentrations between 0 and 50 μg/mL showed no evidence of cytotoxicity to both FLS and RAW264.7 cells (data not shown). FLS in the synovium play a critical role in maintaining inflammation and destroying cartilage by producing pro-inflammatory cytokines [37]. FLS have been reported to be able to express TLR4 as well as a variety of adhesion molecules [37,38]. Macrophages, which are the main source of pro-inflammatory cytokines such as TNF-α and IL-1, also participate in inducing inflammation and bone erosion in patients with RA [39]. The in vitro data from the present study demonstrated that GSPE also has an influence on TLR4 expression in human cells in addition to murine cells.
This study has some limitation that the study was designed to include only three groups; control group, GSPE-untreated CIA group, and GSPE-treated CIA group. Inclusion of GSPE-treated control group would have given more information about basic effect of GSPE on control DBA/1J mice. We chose one dosage (100 mg/kg) of GSPE in the study based on previous study as our study was focused on revealing the new mechanism of action of GSPE [13]. However, different dosage group of GSPE would have made the study more informative. CIA group co-treated with GSPE and TLR4 agonist would have also provided more information that TLR4 inhibition by GSPE could be reversed by TLR4 agonist in CIA mice. GSPE was given intraperitoneally as we intended to administer more accurate dosage of GSPE to each mouse in our study. However, oral administration of GSPE might approach to clinical applications more closely.
In conclusion, our study demonstrated that GSPE attenuated autoimmune arthritis by regulating the TLR4/MyD88/NF-κB signaling pathway. GSPE effectively suppressed anti-type II collagen IgG and pro-inflammatory cytokines. A crucial innate receptor for autoimmune diseases, TLR4 expression in the synovial tissue was significantly influenced by GSPE in vivo, as well as in vitro. The results suggest that GSPE could be effective in treating immunological diseases such as RA, in the pathogenesis of which TLR4 activation is involved.
KEY MESSAGE
1. Our study demonstrated that grape seed proanthocyanidin extract (GSPE) ameliorated collagen-induced arthritis by regulating the Toll-like receptor 4 (TLR4)-MyD88-NF-κB signaling pathway.
2. GSPE also regulated the expression of TLR4 in human fibroblast-like synoviocytes.
3. Further studies would provide more evidence that GSPE could be effective in treating immunological diseases such as rheumatoid arthritis, in the pathogenesis of which TLR4 activation is involved.



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print





