 |
 |
| Korean J Intern Med > Volume 27(2); 2012 > Article |
|
Abstract
In the 1980s, Gram-negative pathogens appeared to have been beaten by oxyimino-cephalosporins, carbapenems, and fluoroquinolones. Yet these pathogens have fought back, aided by their membrane organization, which promotes the exclusion and efflux of antibiotics, and by a remarkable propensity to recruit, transfer, and modify the expression of resistance genes, including those for extended-spectrum ╬▓-lactamases (ESBLs), carbapenemases, aminoglycoside-blocking 16S rRNA methylases, and even a quinolone-modifying variant of an aminoglycoside-modifying enzyme. Gram-negative isolates -both fermenters and non-fermenters-susceptible only to colistin and, more variably, fosfomycin and tigecycline, are encountered with increasing frequency, including in Korea. Some ESBLs and carbapenemases have become associated with strains that have great epidemic potential, spreading across countries and continents; examples include Escherichia coli sequence type (ST)131 with CTX-M-15 ESBL and Klebsiella pneumoniae ST258 with KPC carbapenemases. Both of these high-risk lineages have reached Korea. In other cases, notably New Delhi Metallo carbapenemase, the relevant gene is carried by promiscuous plasmids that readily transfer among strains and species. Unless antibiotic stewardship is reinforced, microbiological diagnosis accelerated, and antibiotic development reinvigorated, there is a real prospect that the antibiotic revolution of the 20th century will crumble.
The ability to treat bacterial infection is critical to modern medicine, from abdominal surgery to transplants to cancer management. Without antibiotics, these procedures would be unthinkable owing to the infection risk. Yet this ability to beat infection has existed for only a brief fraction of human history, beginning with the introduction of sulfonamides in the 1930s. The 'antibiotic revolution' gathered pace from the late 1940s to the 1960s, with a golden age of natural product discovery. This period was followed by two decades of repeated successes in modifying natural antibiotics to enhance their activity. Subsequently, the flow of new antibiotics has dwindled, with diminishing numbers licensed in the 1990s and the first decade of this century [1,2]. The reasons are threefold: first, the technical difficulty of discovering new antibiotics, especially those able to penetrate Gram-negative bacteria [3,4]; second, the regulatory hurdles to new antibiotics which have become increasingly complex [5]; and, third, the brutal fact that antibiotics, as short-course treatments, are less lucrative for the pharmaceutical industry than are long-term therapies for chronic conditions [6,7].
The lack of new antibiotics is critically important, given the continued accumulation of resistance favored by Darwinian selection, which erodes the utility of existing antibiotics. Once a pathogen has been cultured some antibiotic retaining activity can usually be identified, at least in vitro. The greater problem is for empirical therapy, where growing resistance increases the risk that the antibiotic used will prove inactive. In severely ill patients, this ineffectiveness leads to increased mortality, length of hospital stay, and cost [8,9].
This review considers the extent of emerging and proliferating resistance in Gram-negative bacteria-where the problem is most acute-and discusses strategies that may be deployed to counter the threat.
The defining feature of Gram-negative bacteria, distinguishing them from Gram-positive species, is a double membrane wall (Fig. 1). The outer membrane excludes large or hydrophobic molecules, including glycopeptides, daptomycin, and rifampicin, and slows the entry of drugs that can cross it, augmenting the effectiveness of the next line of defense. These defenses include periplasmic-╬▓-lactamases and cytoplasmic membrane efflux pumps, which scavenge amphipathic molecules-including antibiotics-that enter the membrane, pumping them back out of the cell [10]. The combination of impermeability and clearance explains why Gram-negative bacteria are inherently more resistant than Gram-positive species, and why new antibiotics active against them are harder to find [3]. Efflux is especially important in Pseudomonas aeruginosa, which has multiple pumps that render it resistant to many drug families active against other Gram-negative species [11,12].
Both Enterobacteriaceae (Escherichia coli, Klebsiella spp., Enterobacter spp., Proteus spp., Serratia spp.) and non-fermenters (Pseudomonas, Acinetobacter, and related genera) are important opportunist pathogens. E. coli is-by a large margin-the most common agent of community and hospital urinary tract infections and the main aerobic component of the mixed flora in intra-abdominal infections. P. aeruginosa, Klebsiella, and Enterobacter are important in nosocomial pneumonia, especially in patients with prolonged hospitalization. Acinetobacter baumannii is less pathogenic, but notoriously causes outbreaks in the most vulnerable patients in intensive care units (ICUs). Gram-negative pathogens account for around half of all bacteremias, most of which arise due to bacterial overspill from other primary infection sites. In the UK, E. coli is the most common agent of bacteremia, causing 30% of all cases, whereas P. aeruginosa, Klebsiella spp., Enterobacter spp., and Proteeae each account for 4-8% of cases [13]. A few Gram-negative species are important classical pathogens; those remaining important in developed countries include Salmonella spp., Haemophilus influenzae, Moraxella catarrhalis, Neisseria meningitidis, and N. gonorrhoeae. Others ranging from Vibrio cholenzae, to Burkholderia pseudomallei remain important in developing countries, but lie beyond the scope of this review.
The first anti-Gram-negative antibiotics were sulfonamides, introduced in the 1930s; tetracyclines, first-generation aminoglycosides, and chloramphenicol then became available in the late 1940s and early 1950s, respectively. Two quantum leaps followed. The first delivered semi-synthetic penicillins and first-generation cephalosporins in the early 1960s, along with more potent and less toxic aminoglycosides such as gentamicin and amikacin. The second, in the 1980s, led to the development of third- and fourth-generation oxyimino-cephalosporins, ╬▓-lactamase-inhibitor combinations, carbapenems, and fluoroquinolones. These drug families are now standard therapies for severe Gram-negative infections, but growing resistance has created the present problem, as few new agents have been developed to replace them.
At launch, third-generation cephalosporins were almost universally active against Enterobacteriaceae; ceftazidime was also active against P. aeruginosa and, often, A. baumannii. Resistance emerged first in Enterobacter spp. (especially), Citrobacter freundii, and Serratia spp., all of which have inducibly-expressed chromosomal AmpC ╬▓-lactamases. Third-generation cephalosporins are labile to these enzymes but are weak inducers of their synthesis. The result is that cephalosporins kill the AmpC-inducible cells but confer an advantage upon spontaneous 'derepressed' variants that express AmpC activity without induction [14]. These variants arise by spontaneous mutation in inducible (i.e., normal) strains, and the likelihood of their selection during therapy, with consequent treatment failure, is as high as 20-30% when third-generation cephalosporin is used to treat an Enterobacter bacteraemia [15]. Selection is associated with a doubling of mortality and a 50% increase in hospital stay and doubling of cost for survivors (Table 1) [16]. Piperacillin-tazobactam is also compromised, but seems to be less often associated with selection than are third-generation cephalosporins, although the reasons for this difference remain uncertain and one analysis did find similar selectivity [17]. Fourth-generation cephalosporins (cefepime and cefpirome) and carbapenems are more stable and less selective (Table 2).
Genes encoding AmpC ╬▓-lactamases have escaped from the chromosomes of Citrobacter and other genera to plasmids and are now circulating in E. coli and Klebsiella pneumoniae [18]. The enzymes they encode are variously called CMY, FOX, MOX and MIR types or, more usefully, 'plasmid AmpC.' Extended-spectrum ╬▓-lactamases (ESBLs) represent the second and greater plasmid-mediated threat to oxyimino-cephalosporins, affecting fourth- as well as third-generation analogues (Table 2). The earliest ESBLs, first identified in the 1980s, were mutants of the plasmid-borne TEM and SHV penicillinases that had disseminated in E. coli and other Enterobacteriaceae in the 1960s and 1970s [19]. Although third-generation cephalosporins are stable to classical TEM and SHV enzymes, over 200 ESBL mutants have been identified, with sequence changes that expand the active site, allowing attack on oxyimino-cephalosporins (see http://www.lahey.org/studies for a list). Many variants have been described only once or twice, but SHV-2, -5, -11, and -12 and TEM-3, -10, -26, -52, and -116 are widespread in many countries [20,21].
TEM and SHV ESBLs spread during the 1980s and 1990s, largely in Klebsiella spp. and particularly in ICUs [19,22]. Some strains caused major outbreaks, but did not substantially penetrate E. coli strains or spread in the community. This is surprising because E. coli strains with the progenitor TEM-1 enzyme are frequent in the human gut and must have been exposed to huge amounts of ceftriaxone, which was long the most widely used injectable antibiotic and has biliary excretion. These ESBLs may thus present a burden to E. coli, but this aspect remains to be properly explored.
A massive shift in the distribution of ESBLs has occurred since 2000 with the spread of CTX-M types-a decade earlier in Argentina [23]. Unlike TEM and SHV variants, these ESBLS are not mutants of old plasmid-mediated penicillinases. Rather, their genes originated in the chromosomes of Kluyvera spp., a genus of no direct clinical importance, and were mobilized to plasmids by insertion sequences, notably ISEcp1 [24,25]. Plasmids encoding these CTX-M enzymes then reached human opportunists, where they have proliferated in community E. coli and hospital Klebsiella spp. Gene escape has occurred repeatedly, with five CTX-M families (groups 1, 2, 8, 9, and 25) circulating. Different CTX-M families dominate in different places: CTX-M-15 (group 1) is predominant in most of Europe, North America, the Middle East, and India, but CTX-M-14 (group 9) is most common in China, Southeast Asia, and Spain, and CTX-M-2 (group 2) is predominant in Argentina, Israel, and Japan [20,22].
The prevalence of ESBLs (CTX-M and other types) differs among patient groups and clinical and geographic settings. In the UK, around 10% of E. coli from bacteremias now have ESBLs -90% of them CTX-M types [26]- and producers are frequent in urinary isolates from geriatric patients, although denominators are poor, precluding accurate prevalence estimates. By contrast, their prevalence is Ōēż 1% in community cystitis. Gut carriage of ESBL E. coli was seen in 40% of nursing home residents in Belfast (Northern Ireland), providing a reservoir for future urinary or intra-abdominal infections-which are frequent in these vulnerable patients-but in only 1-2% of other outpatients whose stools were examined by the clinical laboratory [27]. Many studies have shown that community infections with ESBL E. coli predominantly affect older (> 65 years) patients with recent hospitalization and antimicrobial treatment, especially with cephalosporins or quinolones [28]. Travel in high-prevalence countries is a risk factor for gut colonization by ESBL E. coli, with travel-associated-strains sometimes causing later urinary infections [29].
The variability in ESBL rates among population groups should prompt caution when viewing national prevalence rates; these benchmark values are of less clinical importance than local rates in different patient groups. Despite this caveat, a rough international picture can be drawn. Multiple surveys have shown that the highest ESBL rates for E. coli and Klebsiella spp. occur in India (Ōēź 80%) and China (Ōēź 60%), [30-32], with rates Ōēź 30% elsewhere in East and Southeast Asia, Latin America, and southern Europe vs. 5-10% in Australasia, northern Europe, and North America [30]. Some studies have suggested that ESBL prevalence in India is equally high in community and hospital E. coli isolates [31], probably reflecting unregulated antibiotic use coupled with recycling of gut bacteria by an overloaded sewage infrastructure [33,34]. In Hong Kong, ESBLs were found in 7.3% of E. coli from uncomplicated community cystitis, including in 11/219 isolates from women aged 18-35 years, a much younger age group than typically affected by ESBL E. coli elsewhere [35]. A growing number of ESBL outbreaks has also been reported in neonatal units, likely due to the transfer of E. coli from the gut flora of one mother to her infant at birth, followed by spread among other infants in the unit [36,37].
The epidemiology of CTX-M ESBLs involves both plasmid dissemination and clonal success. CTX-M-15-the most prevalent enzyme-is most often encoded by IncF plasmids that are prone to multiple rearrangements [38]. In the case of K. pneumoniae, these plasmids are carried by diverse strains; some strains cause local outbreaks but none has achieved wider success [39]. In E. coli, however, CTX-M-15-encoding IncF plasmids are strongly associated with a sequence type (ST)131 lineage, now widespread globally [25,40] (Fig. 2). It remains unclear whether a progenitor ST131 strain acquired an original IncF-CTX-M-15 plasmid and then spread while diversifying, or whether ST131-a lineage that dates back to the 1980s-has repeatedly acquired IncF-CTX-M-15 plasmids at different times in different places, perhaps with considerable exchange to and from Klebsiella. E. coli ST131 occasionally hosts other ESBLs besides CTX-M-15: in Belfast (Northern Ireland, UK) it often has the IncI1 plasmid, pEK203, encoding CTX-M-3. CTX-M-3 differs from CTX-M-15 by only a single amino acid change and the two enzymes can interconvert by mutation: nevertheless, the sequences flanking blaCTX-M-3 in pEK203 differ from those flanking blaCTX-M-15 in its typical IncF plasmid hosts, e.g., pEK499 and pEK516 [41,42], showing that they reflect separate gene escapes from Kluyvera. CTX-M-14 ESBL, which dominates in the Far East, is not strongly associated with ST131 or other successful clones, and its accumulation largely entails plasmid spread among strains.
ESBLs achieved a high prevalence before 2000 in Korea; otherwise, the pattern appears similar to that elsewhere, with CTX-M ESBL types replacing TEM and SHV. In a review of studies published before 1998, Pai [43] estimated ESBL prevalence rates of 4.8-7.5% in E. coli and 22.5-22.8% in K. pneumoniae, with TEM-52 dominant in E. coli and SHV-2 and -12 in K. pneumoniae. Plasmid-mediated CMY-1 (AmpC) was estimated to occur in 15% of Enterobacteriaceae-an exceptionally high rate [43]. These results were broadly supported by a survey of 13 hospitals by Jeong et al. [44] in 2002, which found SHV-2a and -12 and TEM-19, -20, -52, and -116 to be the main ESBLs, although the prevalence of CMY-1 was much lower than Pai [43] had suggested. CTX-M ESBLs were first reported in Korea in 2005 [45] and are now predominant [46]: CTX-M-14 and -15 are both widespread [46,47]. As elsewhere, CTX-M-15 is strongly associated with E. coli ST131 [48], whereas CTX-M-14 is disseminated among strains [47]. Infection risk factors for ESBL producers resemble those found internationally, including prior healthcare and antibiotics [49,50]. More exceptional-but recapitulating the Hong Kong data [35]-is a 2006 multicenter survey finding of ESBLs in 11.8% of E. coli from uncomplicated cystitis [51]. A subsequent study of isolates collected in 2008-2009 found a lower (6.4%) rate of cephalosporin resistance in E. coli from cystitis, although the authors noted that ciprofloxacin resistance continued to increase from 15.2% in 2002 to 23.4% in 2006 and 24.8% in 2008-2009 [52].
Many ESBL producers in Korea and elsewhere are multiresistant to non-╬▓-lactam antibiotics, including fluoroquinolones and aminoglycosides [20,23,53]. The main factors in quinolone resistance are combinations of mutations affecting the chromosomal topoisomerase genes gyrA and parC. Resistance to other antibiotics, including trimethoprim, tetracyclines, sulfonamides, and chloramphenicol as well as aminoglycosides is often encoded by the same plasmids that determine the ESBL. IncF plasmids encoding CTX-M-15 ESBL often also determine AAC(6')-Ib-cr, which inactivates amikacin and tobramycin (not gentamicin) and also deactivates ciprofloxacin, augmenting resistance [54]. These plasmids also often encode OXA-1, an inhibitor-resistant penicillinase, conferring resistance to piperacillin-tazobactam and amoxicillin-clavulanate, even though these inhibitors inactivate CTX-M-15 [41,55].
Most aminoglycoside resistance among Enterobacteriaceae in the West is determined by aminoglycoside-modifying enzymes like AAC(6')-1b. These enzymes are also common in Asia, but wide circulation of the ArmA and RmtA-C 16S ribosomal RNA (rRNA) methylases is also found in Asia, unlike in Europe and the US [56-58]. These methylases modify bacterial ribosomes to block binding of all key aminoglycosides, including the new analogue ACHN-490 [59]. They are widespread in Korea and are commonly carried by plasmids that also encode SHV-12 and CTX-M-14 ESBLs, but have not been associated with CTX-M-15 or E. coli ST131 [60,61].
The growing prevalence of cephalosporin-resistant Enterobacteriaceae is driving the directed and empirical use of carbapenems, as the only widely marketed ╬▓-lactams stable to ESBLs and AmpC enzymes (Table 2).
Carbapenems retained near-universal activity against Enterobacteriaceae for around 20 years after the introduction of imipenem in 1985, but resistance is now accumulating. It can evolve by porin loss, reducing drug entry in strains already containing high levels of AmpC or an ESBL [62]. This is a common source of low-level resistance to ertapenem, especially in Enterobacter and Klebsiella, with resistant variants sometimes selected during carbapenem therapy [63]. However, this problem is limited because these variants are often unstable and tend to revert to susceptibility, rarely spreading among patients [64,65]. Their porin deficiency likely impedes nutrition, reducing competitiveness.
Much more concerning is the gradual proliferation of carbapenemases, which are biochemically diverse and include members of three of the four ╬▓-lactamase classes (A, B, D).
KPC enzymes (class A, non-metallo) are currently the predominant carbapenemases emerging in Enterobacteriaceae [66]. They were first recorded in North Carolina (USA) in 1997, with outbreaks in New York by 2005. Since 2006, KPC+
K. pneumoniae have spread across the USA and in Israel, Greece, and, latterly, Italy, with growing outbreaks in China, Brazil, and several European countries. The highest national prevalence rate (to 40%) is for K. pneumoniae from bacteremias in Greece [67]. Much of this spread reflects an association of KPC enzymes with a successful clone, K. pneumoniae, ST258 (Fig. 3) [68]. Members of this lineage are typically resistant to all antibiotics except colistin, tigecycline, and gentamicin. A colistin-resistant variant of ST258 is circulating in Greece [69], with cases also seen, possibly via import, in Hungary and the UK [70]. The reasons for the particular success of ST258 are uncertain, but its ability to spread swiftly is beyond dispute. In addition, KPC enzymes have spread considerably via plasmids among different K. pneumoniae strains and other Enterobacteriaceae, principally E. coli and Enterobacter spp.
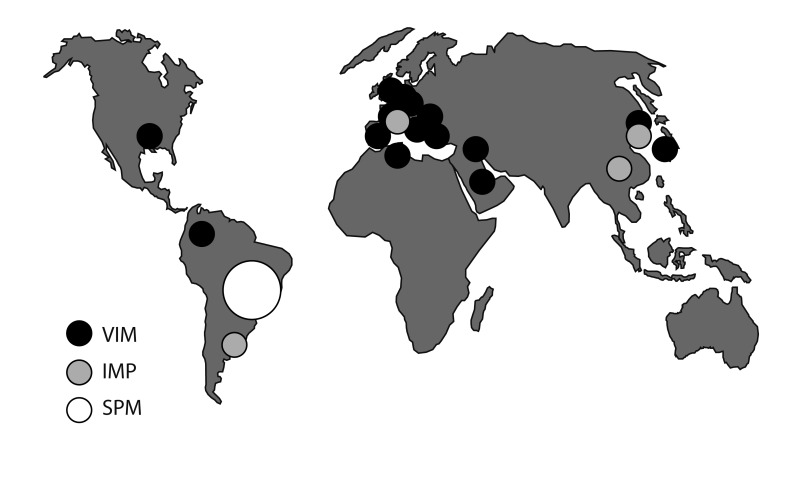
Metallo-carbapenemases (class B) are biochemically very different from KPC, and include IMP, VIM, and New Delhi Metallo (NDM) enzymes, along with less frequently occurring families. IMP was recognized in Japan at the end of the 1980s [71] and has gradually spread worldwide, although it has not achieved great prevalence anywhere. VIM is more important; it was first recorded in P. aeruginosa in Verona (Italy) in 1997 [72] and then spread into Enterobacteriaceae, principally K. pneumoniae. By 2005, it was present in 40% of bloodstream K. pneumoniae from Greece, where it has subsequently been partly displaced by KPC enzymes [73].
Clusters of K. pneumoniae with VIM carbapenemases are increasingly being reported worldwide, although they are less numerous than isolates with KPC enzymes. A further distinction is that VIM enzymes are not associated with any particular clone, whereas KPC enzymes are associated with ST258 K. pneumoniae. Although local outbreaks occur, the larger issue is the dissemination of blaVIM among K. pneumoniae, largely by IncN plasmids [73,74].
NDM-the most publicized metallo-carbapenemase-is epicentered in India, where it occurs in 2-8% of Enterobacteriaceae at teaching hospitals [75,76], and in Pakistan, where 27% of inpatients at two military hospitals were carriers. blaNDM-1, encoding the enzyme, is wide-spread in surface-water bacteria in New Delhi, implying community circulation. Enterobacteriaceae (and occasionally Acinetobacter spp.) with NDM carbapenemase have repeatedly been imported into Europe, Asia, North America, and Australasia by those who have travelled or been hospitalized in the Indian subcontinent [77]. As with VIM metallo-carbapenemases, spread is largely by plasmid transfer, not clonal dissemination [77].
The final and very different-emerging carbapenemase of Enterobacteriaceae is OXA-48, reported in Turkey since 2001, mostly in K. pneumoniae. It has disseminated in the Middle East and North Africa since 2007-2008, with repeated import into Europe; outbreaks have occurred in the UK, Belgium, Ireland, France, Spain, and The Netherlands (Fig. 4) [78,79]. The main factor in its spread is dissemination of 50- and 70-kb plasmids carrying the blaOXA-48 transposon Tn1999 among Klebsiella strains [80]. A related enzyme, OXA-181, is circulating in India, where it seems less frequent than NDM-1 [81]. OXA-48 and -181 originated by separate mobilizations of chromosomal ╬▓-lactamase genes from Shewenella, a genus otherwise lacking clinical importance [79].
To date, carbapenemase-producing Enterobacteriaceae have shown little circulation in Korea, although C. freundii and S. marcescens with VIM-2 [82] and K. pneumoniae [83] with KPC enzymes have been reported, and a very recent report described an outbreak cluster of K. pneumoniae with the NDM-1 enzyme [84]. Unusually, an S. marcescens strain with AmpC and impermeability caused an outbreak involving nine patients at a hospital in Seoul [65]. Most disturbing is the recent appearance of KPC+
K. pneumoniae ST258. Only one producer has been reported to date [85], from a urinary infection in Seoul, but this development should raise concern, given the strain's dramatic success elsewhere.
KPC carbapenemases inactivate all ╬▓-lactams. Aztreonam is stable to the IMP, VIM, and NDM types, but these are often accompanied by aztreonam-hydrolyzing ESBLs. Similarly, although OXA-48 spares ceftazidime, it is often accompanied by ceftazidime-hydrolyzing ESBLs (Table 2). Resistance to non-╬▓-lactams is variable, but many carbapenemase producers are susceptible only to colistin, tigecycline, and/or fosfomycin, none of which is an 'ideal' antibiotic. NDM carbapenemases are often accompanied by the 16S rRNA methylases mentioned earlier, compromising the widest range of aminoglycosides [59,81].
The prevalence of resistance in P. aeruginosa varies widely among countries. In Europe, resistance rates to aminoglycosides, carbapenems, cephalosporins, and fluoroquinolones are lowest (< 10%) in the northwest, particularly in Scandinavia, and highest (25-50%) in the southeast, particularly in Greece [86]. Rates are high in Latin America and Southeast Asia, and intermediate in the US [87].
Most resistance among P. aeruginosa in Europe and North America involves mutations that restrict drug entry (the most common cause of carbapenem resistance), derepress chromosomal AmpC (as in Enterobacter, above) and up-regulate efflux [88,89]. Acquired mechanisms are more important in resistance in Asia and Latin America and include different ESBLs to those most prevalent in Enterobacteriaceae, notably PER-1, which is widespread in Turkey [90], and VEB-1, which first proliferated in Southeast Asia [91]. Metallo-╬▓-lactamases are scattered and single-hospital outbreaks due to producers have occurred worldwide (Fig. 5) [92,93]. These outbreaks are often prolonged, with cases scattered over long periods, probably owing to persistence in the hospital structure, e.g., in sink traps. In Brazil, a clone with S├Żo Paolo Metallo (SPM)-╬▓-lactamase has disseminated among hospitals, achieving national distribution [94]. Elsewhere, P. aeruginosa ST111, which corresponds to serotype O12 and 'burst group 4,' is now a common host to VIM metallo-carbapenemases [68]. This lineage has a long history of acquiring resistance -20 years ago it often carried the PSE-1 carbenicillinase [95]. ST235 (serotype 11) is also a successful accumulator of resistance, frequently hosting IMP and VIM carbapenemases [68].
A 2005 survey of 23 hospitals in Korea found carbapenemases in 10% of carbapenem-resistant P. aeruginosa, with VIM types 2.5-fold more common than IMP types [96]. The remaining 90% of carbapenem-resistant P. aeruginosa were inferred to have impermeability-type resistance. Subsequently, an ST235 clone with the IMP-6 enzyme has disseminated nationally [97]. In a 2009 survey of 21 hospitals, this strain accounted for 30/386 (7.8%) isolates and was recovered in 14 sites. As with NDM-1 in Enterobacteriaceae, blaIMP-6 in Korean P. aeruginosa is often associated with the ArmA rRNA methylase [98].
The molecular epidemiology of A. baumannii, another important opportunistic non-fermenter, contrasts to that of P. aeruginosa, with resistance largely originating from the up-regulation of inherent chromosomal AmpC and carbapenemase (blaOXA-51) genes by insertion sequences, critically ISAba1, which migrate within the chromosome [99,100]. The insertion of ISAba1 in front of one of these ╬▓-lactamase genes provides an efficient promoter, allowing expression and resistance. Alternatively, resistance can be caused by acquired OXA-23, -40, and -58 carbapenemases [99]. OXA-23 originated as a chromosomal carbapenemase in Acinetobacter radioresistens [101], a species with no direct clinical importance.
OXA carbapenemases account for most carbapenem resistance in A. baumannii worldwide, including in Korea, where the resistant proportion reportedly grew from 13% in 2003 to 51% in 2008-2009, and where two carbapenemase-producing clones are in wide circulation among hospitals, one with OXA-23 and frequent colistin resistance, the other with up-regulated OXA-51 [102].
Metallo-carbapenemases also occur, with NDM recorded in A. baumannii (although not yet in Korea) and IMP in non-baumannii species, but these are extremely rare compared with OXA types [103]. A unique metallo-type, Seoul imipenemase (SIM-1), has been described from A. baumannii in Korea, but it has not disseminated [104].
As in Enterobacteriaceae, most resistance to non-╬▓-lactams depends on acquired genes-not on mutation, as in P. aeruginosa. Acquired genes may be carried by plasmids or by massive 'resistance islands' inserted into the chromosome [105,106]. Colistin resistance, as in one of the prevalent Korean clones, can arise via changes to membrane phospholipids and lipopolysaccharides [107,108], and resistance to tigecycline by up-regulation of the chromosomal AdeB efflux pump is sometimes selected for during therapy [109].
A few N. meningitidis and H. influenzae isolates have low-level resistance to fluoroquinolones, whereas resistance to third-generation cephalosporins has not been seen in either species. N. meningitidis shows only slightly reduced susceptibility to penicillin, even after 70 years of use.
N. gonorrhoeae, by contrast, has sequentially developed resistance to low-level penicillin, high-level penicillin, and ciprofloxacin, with borderline non-susceptibility to cefixime (MICs, 0.12-0.5 mg/L) now proliferating [110], including in Korea [111,112]. This development is driving a treatment shift to high-dose ceftriaxone, sometimes (as in the UK) combined with azithromycin [113]. Few other options are available if substantial resistance to these treatments emerges and, disturbingly, two reports have described gonococci with resistance to ceftriaxone and cefixime: one from Japan [114] and the other from France [115]. The first patient developed an oropharyngeal infection and recovered-perhaps spontaneously-during a second course of ceftriaxone; the second developed a urethral infection that was cured with gentamicin.
The range of antibiotic options to treat Gram-positive infections, particularly those due to MRSA, has expanded in the past decade with the introduction of quinupristin-dalfopristin, linezolid, daptomycin, tigecycline, and, in some markets, telavancin and ceftaroline. Additional agents, including oritavancin, dalbavancin, and tedizolid (torezolid), are in phase III development and are anticipated in the next 2-3 years. Among these new anti-MRSA agents, only tigecycline shows any increase in in vitro anti-Gram-negative activity compared with previous members of its chemical family (tetracyclines), and its efficacy in severe infections is variable, with low serum levels and disappointing results in nosocomial pneumonia [116]. The only other anti-Gram-negative agent launched in the past decade is doripenem, which represents an incremental improvement over meropenem in anti-Pseudomonas activity, but is otherwise similar [117].
In short, little progress against Gram-negative bacteria has been made for a long time. Nevertheless, several new anti-Gram-negative agents are in phase III development and show promise. CXA-201, a cephalosorin (ceftolozane)-tazobactam combination, is four-fold more active than ceftazidime-still the best anti-pseudomonal cephalosporin-against P. aeruginosa [118,119]. Ceftazidime-avibactam, another new inhibitor combination, is active against strains with AmpC, ESBLs, and KPC or OXA-48 carbapenemases [120], and the neoglycoside plazomicin overcomes nearly all aminoglycoside-modifying-enzyme-mediated resistance [121].
Nevertheless, each of these agents has gaps in its spectrum, and none provides such complete coverage as ceftazidime, imipenem, and ciprofloxacin did when they were introduced 20-30 years ago. CXA-201 lacks activity against Enterobacteriaceae with metallo- and KPC carbapenemases and shows borderline activity against some ESBL and AmpC producers [122]; ceftazidime-avibactam lacks activity against strains with metallo-carbapenemases, such as VIM and NDM [120]; and plazomicin fails to overcome resistance mediated by ArmA and Rmt rRNA methylases [59].
Other antibiotics in earlier developmental stages may offer a more comprehensive solution. The most promising are 1) boron-based protein synthesis inhibitors (GSK-Anacor partnership) [123]-though their development was recently suspended, 2) fluorine-containing tetracyclines ('fluorocyclines,' Tetraphase) [124], and 3) novel protein synthesis inhibitors (RibX-Sanofi partnership). Aztreonam-avibactam shows the potential to overcome both metallo- and non-metallo-carbapenemases [120]; it is not yet under formal development but would be relatively easy to progress, following experience with ceftazidime-avibactam and ceftaroline-avibactam.
However, these drugs will only be available in the future, and licensing success is not guaranteed. Therefore, as much as possible must be done through good stewardship and infection control to prolong the life of existing antibiotics. Improvements in laboratory microbiology and its interaction with patient management are also critical. Currently, a 48-hour interval-at the very best-remains between the clinical diagnosis of a patient's infection and the clinician's acquisition of laboratory results indicating the isolate's susceptibility. During this interval the patient is treated empirically based on the likely pathogens and local resistance rates. Such treatment becomes more difficult as resistance increases, and multiple studies have shown that resistance to the empirical regimen leads to a near doubling of mortality in bacteremias [8]. One response is to start patients on the most powerful empirical antibiotic, typically a carbapenem combined with an anti-MRSA agent, and to 'de-escalate' once laboratory microbiology results are available [125,126]. Although this strategy improves initial coverage, which may benefit the patient [127], it has two pitfalls: first, hospitals frequently fail to enforce de-escalation when a susceptible pathogen is isolated; second, the approach does not specify the appropriate response when the patient still shows symptoms of infection but no pathogen is isolated. Both factors potentially lead to unnecessarily prolonged use of the broadest-spectrum agents, increasing the selection of resistance. The situation would be improved if microbiology could be accelerated, identifying pathogens and their resistances in hours rather than days, and without the need for culture [128]. Potential methods exist, including polymerase chain reaction or DNA arrays to seek species- and resistance-specific genes [129,130]. Nevertheless, great challenges remain, especially with non-sterile-site specimens, in which DNA extracted from the pathogen becomes mixed with that from commensals.
If these challenges can be overcome-perhaps first for blood and other sterile-site specimens-results can be delivered to the ward far more swiftly. Such development would reduce the conflict between the individual, who currently may benefit from receiving the most powerful antibiotics, and society, which benefits if they are conserved. This goal is surely worthwhile because the alternative is a world where high-tech medicine becomes ever more hazardous as-owing to proliferating resistance-it becomes increasingly more difficult to treat infection empirically.
References
1. Boucher HW, Talbot GH, Bradley JS, et al. Bad bugs, no drugs: no ESKAPE! An update from the Infectious Diseases Society of America. Clin Infect Dis 2009;48:1ŌĆō12PMID : 19035777.


2. Holmes NE, Howden BP. The rise of antimicrobial resistance: a clear and present danger. Expert Rev Anti Infect Ther 2011;9:645ŌĆō648PMID : 21692668.


3. Livermore DM. British Society for Antimicrobial Chemotherapy Working Party on The Urgent Need: Regenerating Antibacterial Drug Discovery and Development. Discovery research: the scientific challenge of finding new antibiotics. J Antimicrob Chemother 2011;66:1941ŌĆō1944PMID : 21700626.


4. Payne DJ, Gwynn MN, Holmes DJ, Pompliano DL. Drugs for bad bugs: confronting the challenges of antibacterial discovery. Nat Rev Drug Discov 2007;6:29ŌĆō40PMID : 17159923.


5. Finch R. BSAC Working Party on the Urgent Need: Regenerating Antibacterial Drug Discovery and Development. Regulatory opportunities to encourage technology solutions to antibacterial drug resistance. J Antimicrob Chemother 2011;66:1945ŌĆō1947PMID : 21700629.


6. White AR. BSAC Working Party on the Urgent Need: Regenerating Antibacterial Drug Discovery and Development. Effective antibacterials: at what cost? The economics of antibacterial resistance and its control. J Antimicrob Chemother 2011;66:1948ŌĆō1953PMID : 21700625.


7. Projan SJ. Why is big pharma getting out of antibacterial drug discovery? Curr Opin Microbiol 2003;6:427ŌĆō430PMID : 14572532.


8. Paul M, Shani V, Muchtar E, Kariv G, Robenshtok E, Leibovici L. Systematic review and meta-analysis of the efficacy of appropriate empiric antibiotic therapy for sepsis. Antimicrob Agents Chemother 2010;54:4851ŌĆō4863PMID : 20733044.



9. Kollef MH. Broad-spectrum antimicrobials and the treatment of serious bacterial infections: getting it right up front. Clin Infect Dis 2008;47(Suppl 1):S3ŌĆōS13PMID : 18713047.


10. Li XZ, Nikaido H. Efflux-mediated drug resistance in bacteria: an update. Drugs 2009;69:1555ŌĆō1623PMID : 19678712.



11. Alvarez-Ortega C, Wiegand I, Olivares J, Hancock RE, Martinez JL. The intrinsic resistome of Pseudomonas aeruginosa to ╬▓-lactams. Virulence 2011;2:144ŌĆō146PMID : 21304266.


12. Pages JM, Amaral L. Mechanisms of drug efflux and strategies to combat them: challenging the efflux pump of Gram-negative bacteria. Biochim Biophys Acta 2009;1794:826ŌĆō833PMID : 19150515.


13. Wilson J, Elgohari S, Livermore DM, et al. Trends among pathogens reported as causing bacteraemia in England, 2004-2008. Clin Microbiol Infect 2011;17:451ŌĆō458PMID : 20491834.


14. Livermore DM. Clinical significance of beta-lactamase induction and stable derepression in gram-negative rods. Eur J Clin Microbiol 1987;6:439ŌĆō445PMID : 3311738.


15. Kaye KS, Cosgrove S, Harris A, Eliopoulos GM, Carmeli Y. Risk factors for emergence of resistance to broad-spectrum cephalosporins among Enterobacter spp. Antimicrob Agents Chemother 2001;45:2628ŌĆō2630PMID : 11502540.



16. Cosgrove SE, Kaye KS, Eliopoulous GM, Carmeli Y. Health and economic outcomes of the emergence of third-generation cephalosporin resistance in Enterobacter species. Arch Intern Med 2002;162:185ŌĆō190PMID : 11802752.


17. Schwaber MJ, Graham CS, Sands BE, Gold HS, Carmeli Y. Treatment with a broad-spectrum cephalosporin versus piperacillin-tazobactam and the risk for isolation of broad-spectrum cephalosporin-resistant Enterobacter species. Antimicrob Agents Chemother 2003;47:1882ŌĆō1886PMID : 12760862.



19. Jacoby GA. Extended-spectrum beta-lactamases and other enzymes providing resistance to oxyimino-beta-lactams. Infect Dis Clin North Am 1997;11:875ŌĆō887PMID : 9421705.


20. Hawkey PM. Prevalence and clonality of extended-spectrum beta-lactamases in Asia. Clin Microbiol Infect 2008;14(Suppl 1):159ŌĆō165PMID : 18154540.


21. Paterson DL, Bonomo RA. Extended-spectrum beta-lactamases: a clinical update. Clin Microbiol Rev 2005;18:657ŌĆō686PMID : 16223952.



22. Bush K. Extended-spectrum beta-lactamases in North America, 1987-2006. Clin Microbiol Infect 2008;14(Suppl 1):134ŌĆō143PMID : 18154537.


23. Livermore DM, Canton R, Gniadkowski M, et al. CTX-M: changing the face of ESBLs in Europe. J Antimicrob Chemother 2007;59:165ŌĆō174PMID : 17158117.


24. Rossolini GM, D'Andrea MM, Mugnaioli C. The spread of CTX-M-type extended-spectrum beta-lactamases. Clin Microbiol Infect 2008;14(Suppl 1):33ŌĆō41PMID : 18154526.


25. Naseer U, Sundsfjord A. The CTX-M conundrum: dissemination of plasmids and Escherichia coli clones. Microb Drug Resist 2011;17:83ŌĆō97PMID : 21281129.


26. Livermore DM. Fourteen years in resistance. Int J Antimicrob Agents 2012;39:283ŌĆō294PMID : 22386741.


27. Rooney PJ, O'Leary MC, Loughrey AC, et al. Nursing homes as a reservoir of extended-spectrum beta-lactamase (ESBL)-producing ciprofloxacin-resistant Escherichia coli. J Antimicrob Chemother 2009;64:635ŌĆō641PMID : 19549667.


28. Rodriguez-Bano J, Picon E, Gijon P, et al. Risk factors and prognosis of nosocomial bloodstream infections caused by extended-spectrum-beta-lactamase-producing Escherichia coli. J Clin Microbiol 2010;48:1726ŌĆō1731PMID : 20181897.



29. Pitout JD, Campbell L, Church DL, Gregson DB, Laupland KB. Molecular characteristics of travel-related extended-spectrum-beta-lactamase-producing Escherichia coli isolates from the Calgary Health Region. Antimicrob Agents Chemother 2009;53:2539ŌĆō2543PMID : 19364876.



30. Hoban DJ, Nicolle LE, Hawser S, Bouchillon S, Badal R. Antimicrobial susceptibility of global inpatient urinary tract isolates of Escherichia coli: results from the Study for Monitoring Antimicrobial Resistance Trends (SMART) program: 2009-2010. Diagn Microbiol Infect Dis 2011;70:507ŌĆō511PMID : 21767706.


31. Chaudhuri BN, Rodrigues C, Balaji V, et al. Incidence of ESBL producers amongst Gram-negative bacilli isolated from intra-abdominal infections across India (based on SMART study, 2007 data). J Assoc Physicians India 2011;59:287ŌĆō292PMID : 21751604.

32. Hsueh PR, Badal RE, Hawser SP, et al. Epidemiology and antimicrobial susceptibility profiles of aerobic and facultative Gram-negative bacilli isolated from patients with intra-abdominal infections in the Asia-Pacific region: 2008 results from SMART (Study for Monitoring Antimicrobial Resistance Trends). Int J Antimicrob Agents 2010;36:408ŌĆō414PMID : 20728316.


33. Tangden T, Cars O, Melhus A, Lowdin E. Foreign travel is a major risk factor for colonization with Escherichia coli producing CTX-M-type extended-spectrum beta-lactamases: a prospective study with Swedish volunteers. Antimicrob Agents Chemother 2010;54:3564ŌĆō3568PMID : 20547788.



34. Walsh TR, Weeks J, Livermore DM, Toleman MA. Dissemination of NDM-1 positive bacteria in the New Delhi environment and its implications for human health: an environmental point prevalence study. Lancet Infect Dis 2011;11:355ŌĆō362PMID : 21478057.


35. Ho PL, Yip KS, Chow KH, Lo JY, Que TL, Yuen KY. Antimicrobial resistance among uropathogens that cause acute uncomplicated cystitis in women in Hong Kong: a prospective multicenter study in 2006 to 2008. Diagn Microbiol Infect Dis 2010;66:87ŌĆō93PMID : 19446980.


36. Velasco C, Rodriguez-Bano J, Garcia L, et al. Eradication of an extensive outbreak in a neonatal unit caused by two sequential Klebsiella pneumoniae clones harbouring related plasmids encoding an extended-spectrum beta-lactamase. J Hosp Infect 2009;73:157ŌĆō163PMID : 19716201.


37. Chandel DS, Johnson JA, Chaudhry R, et al. Extended-spectrum beta-lactamase-producing Gram-negative bacteria causing neonatal sepsis in India in rural and urban settings. J Med Microbiol 2011;60(Pt 4):500ŌĆō507PMID : 21183602.



38. Doumith M, Dhanji H, Ellington MJ, Hawkey P, Woodford N. Characterization of plasmids encoding extended-spectrum ╬▓-lactamases and their addiction systems circulating among Escherichia coli clinical isolates in the UK. J Antimicrob Chemother 2012;67:878ŌĆō885PMID : 22210753.


39. Partridge SR, Zong Z, Iredell JR. Recombination in IS26 and Tn2 in the evolution of multiresistance regions carrying blaCTX-M-15 on conjugative IncF plasmids from Escherichia coli. Antimicrob Agents Chemother 2011;55:4971ŌĆō4978PMID : 21859935.



40. Rogers BA, Sidjabat HE, Paterson DL. Escherichia coli O25b-ST131: a pandemic, multiresistant, community-associated strain. J Antimicrob Chemother 2011;66:1ŌĆō14PMID : 21081548.


41. Woodford N, Carattoli A, Karisik E, Underwood A, Ellington MJ, Livermore DM. Complete nucleotide sequences of plasmids pEK204, pEK499, and pEK516, encoding CTX-M enzymes in three major Escherichia coli lineages from the United Kingdom, all belonging to the international O25:H4-ST131 clone. Antimicrob Agents Chemother 2009;53:4472ŌĆō4482PMID : 19687243.



42. Dhanji H, Doumith M, Rooney PJ, et al. Molecular epidemiology of fluoroquinolone-resistant ST131 Escherichia coli producing CTX-M extended-spectrum beta-lactamases in nursing homes in Belfast, UK. J Antimicrob Chemother 2011;66:297ŌĆō303PMID : 21131323.


43. Pai H. The characteristics of extended-spectrum beta-lactamases in Korean isolates of Enterobacteriaceae. Yonsei Med J 1998;39:514ŌĆō519PMID : 10097677.


44. Jeong SH, Bae IK, Lee JH, et al. Molecular characterization of extended-spectrum beta-lactamases produced by clinical isolates of Klebsiella pneumoniae and Escherichia coli from a Korean nationwide survey. J Clin Microbiol 2004;42:2902ŌĆō2906PMID : 15243036.



45. Jeong SH, Bae IK, Kwon SB, et al. Dissemination of transferable CTX-M-type extended-spectrum beta-lactamase-producing Escherichia coli in Korea. J Appl Microbiol 2005;98:921ŌĆō927PMID : 15752339.


46. Kang CI, Wi YM, Lee MY, et al. Epidemiology and risk factors of community onset infections caused by extended-spectrum ╬▓-lactamase-producing Escherichia coli strains. J Clin Microbiol 2012;50:312ŌĆō317PMID : 22162561.



47. Shin J, Kim DH, Ko KS. Comparison of CTX-M-14- and CTX-M-15-producing Escherichia coli and Klebsiella pneumoniae isolates from patients with bacteremia. J Infect 2011;63:39ŌĆō47PMID : 21712135.


48. Lee MY, Choi HJ, Choi JY, et al. Dissemination of ST131 and ST393 community-onset, ciprofloxacin-resistant Escherichia coli clones causing urinary tract infections in Korea. J Infect 2010;60:146ŌĆō153PMID : 19932131.


49. Park SH, Choi SM, Lee DG, et al. Emergence of extended-spectrum ╬▓-lactamase-producing escherichia coli as a cause of community-onset bacteremia in South Korea: risk factors and clinical outcomes. Microb Drug Resist 2011;17:537ŌĆō544PMID : 21875342.


50. Lee DS, Lee CB, Lee SJ. Prevalence and risk factors for extended spectrum beta-lactamase-producing uropathogens in patients with urinary tract infection. Korean J Urol 2010;51:492ŌĆō497PMID : 20664784.



51. Kim ME, Ha US, Cho YH. Prevalence of antimicrobial resistance among uropathogens causing acute uncomplicated cystitis in female outpatients in South Korea: a multicentre study in 2006. Int J Antimicrob Agents 2008;31(Suppl 1):S15ŌĆōS18PMID : 18096373.


52. Lee SJ, Lee DS, Choe HS, et al. Antimicrobial resistance in community-acquired urinary tract infections: results from the Korean Antimicrobial Resistance Monitoring System. J Infect Chemother 2011;17:440ŌĆō446PMID : 21140281.


53. Paterson DL. Resistance in gram-negative bacteria: Enterobacteriaceae. Am J Infect Control 2006;34(5 Suppl 1):S20ŌĆōS28PMID : 16813978.


54. Karisik E, Ellington MJ, Pike R, Warren RE, Livermore DM, Woodford N. Molecular characterization of plasmids encoding CTX-M-15 beta-lactamases from Escherichia coli strains in the United Kingdom. J Antimicrob Chemother 2006;58:665ŌĆō668PMID : 16870648.


55. Boyd DA, Tyler S, Christianson S, et al. Complete nucleotide sequence of a 92-kilobase plasmid harboring the CTX-M-15 extended-spectrum beta-lactamase involved in an outbreak in long-term-care facilities in Toronto, Canada. Antimicrob Agents Chemother 2004;48:3758ŌĆō3764PMID : 15388431.



56. Fritsche TR, Castanheira M, Miller GH, Jones RN, Armstrong ES. Detection of methyltransferases conferring high-level resistance to aminoglycosides in enterobacteriaceae from Europe, North America, and Latin America. Antimicrob Agents Chemother 2008;52:1843ŌĆō1845PMID : 18347105.



57. Yang J, Ye L, Wang W, Luo Y, Zhang Y, Han L. Diverse prevalence of 16S rRNA methylase genes armA and rmtB amongst clinical multidrug-resistant Escherichia coli and Klebsiella pneumoniae isolates. Int J Antimicrob Agents 2011;38:348ŌĆō351PMID : 21724374.


58. Yu F, Wang L, Pan J, et al. Prevalence of 16S rRNA methylase genes in Klebsiella pneumoniae isolates from a Chinese teaching hospital: coexistence of rmtB and armA genes in the same isolate. Diagn Microbiol Infect Dis 2009;64:57ŌĆō63PMID : 19232867.


59. Livermore DM, Mushtaq S, Warner M, et al. Activity of aminoglycosides, including ACHN-490, against carbapenem-resistant Enterobacteriaceae isolates. J Antimicrob Chemother 2011;66:48ŌĆō53PMID : 21078604.


60. Kang HY, Kim J, Seol SY, Lee YC, Lee JC, Cho DT. Characterization of conjugative plasmids carrying antibiotic resistance genes encoding 16S rRNA methylase, extended-spectrum beta-lactamase, and/or plasmid-mediated AmpC beta-lactamase. J Microbiol 2009;47:68ŌĆō75PMID : 19229493.


61. Kim MH, Sung JY, Park JW, Kwon GC, Koo SH. Coproduction of qnrB and armA from extended-spectrum beta-lactamase-producing Klebsiella pneumoniae. Korean J Lab Med 2007;27:428ŌĆō436PMID : 18160833.


62. Doumith M, Ellington MJ, Livermore DM, Woodford N. Molecular mechanisms disrupting porin expression in ertapenem-resistant Klebsiella and Enterobacter spp. clinical isolates from the UK. J Antimicrob Chemother 2009;63:659ŌĆō667PMID : 19233898.


63. Elliott E, Brink AJ, van Greune J, et al. In vivo development of ertapenem resistance in a patient with pneumonia caused by Klebsiella pneumoniae with an extended-spectrum beta-lactamase. Clin Infect Dis 2006;42:e95ŌĆōe98PMID : 16652304.


64. Garcia-Fernandez A, Miriagou V, Papagiannitsis CC, et al. An ertapenem-resistant extended-spectrum-beta-lactamase-producing Klebsiella pneumoniae clone carries a novel OmpK36 porin variant. Antimicrob Agents Chemother 2010;54:4178ŌĆō4184PMID : 20660683.



65. Suh B, Bae IK, Kim J, Jeong SH, Yong D, Lee K. Outbreak of meropenem-resistant Serratia marcescens comediated by chromosomal AmpC beta-lactamase overproduction and outer membrane protein loss. Antimicrob Agents Chemother 2010;54:5057ŌĆō5061PMID : 20876374.



66. Nordmann P, Cuzon G, Naas T. The real threat of Klebsiella pneumoniae carbapenemase-producing bacteria. Lancet Infect Dis 2009;9:228ŌĆō236PMID : 19324295.


67. Giakkoupi P, Papagiannitsis CC, Miriagou V, et al. An update of the evolving epidemic of blaKPC-2-carrying Klebsiella pneumoniae in Greece (2009-10). J Antimicrob Chemother 2011;66:1510ŌĆō1513PMID : 21543359.


68. Woodford N, Turton JF, Livermore DM. Multiresistant Gram-negative bacteria: the role of high-risk clones in the dissemination of antibiotic resistance. FEMS Microbiol Rev 2011;35:736ŌĆō755PMID : 21303394.


69. Kontopoulou K, Protonotariou E, Vasilakos K, et al. Hospital outbreak caused by Klebsiella pneumoniae producing KPC-2 beta-lactamase resistant to colistin. J Hosp Infect 2010;76:70ŌĆō73PMID : 20705205.


70. Toth A, Damjanova I, Puskas E, et al. Emergence of a colistin-resistant KPC-2-producing Klebsiella pneumoniae ST258 clone in Hungary. Eur J Clin Microbiol Infect Dis 2010;29:765ŌĆō769PMID : 20401676.


71. Watanabe M, Iyobe S, Inoue M, Mitsuhashi S. Transferable imipenem resistance in Pseudomonas aeruginosa. Antimicrob Agents Chemother 1991;35:147ŌĆō151PMID : 1901695.



72. Lauretti L, Riccio ML, Mazzariol A, et al. Cloning and characterization of blaVIM, a new integron-borne metallo-beta-lactamase gene from a Pseudomonas aeruginosa clinical isolate. Antimicrob Agents Chemother 1999;43:1584ŌĆō1590PMID : 10390207.



73. Vatopoulos A. High rates of metallo-beta-lactamase-producing Klebsiella pneumoniae in Greece: a review of the current evidence. Euro Surveill 2008;13:pii: 8023.

74. Garcia-Fernandez A, Villa L, Moodley A, et al. Multilocus sequence typing of IncN plasmids. J Antimicrob Chemother 2011;66:1987ŌĆō1991PMID : 21653604.


75. Lascols C, Hackel M, Marshall SH, et al. Increasing prevalence and dissemination of NDM-1 metallo-╬▓-lactamase in India: data from the SMART study (2009). J Antimicrob Chemother 2011;66:1992ŌĆō1997PMID : 21676902.



76. Deshpande P, Shetty A, Kapadia F, Hedge A, Soman R, Rodrigues C. New Delhi metallo 1: have carbapenems met their doom? Clin Infect Dis 2010;51:1222. PMID : 20964525.

77. Kumarasamy KK, Toleman MA, Walsh TR, et al. Emergence of a new antibiotic resistance mechanism in India, Pakistan, and the UK: a molecular, biological, and epidemiological study. Lancet Infect Dis 2010;10:597ŌĆō602PMID : 20705517.



78. Carrer A, Poirel L, Yilmaz M, et al. Spread of OX A-48-encoding plasmid in Turkey and beyond. Antimicrob Agents Chemother 2010;54:1369ŌĆō1373PMID : 20086157.



79. Potron A, Kalpoe J, Poirel L, Nordmann P. European dissemina tion of a single OXA-48-producing Klebsiella pneumoniae clone. Clin Microbiol Infect 2011;17:E24ŌĆōE26PMID : 21973185.


80. Poirel L, Bonnin RA, Nordmann P. Genetic features of the widespread plasmid coding for the carbapenemase OXA-48. Antimicrob Agents Chemother 2012;56:559ŌĆō562PMID : 22083465.



81. Castanheira M, Deshpande LM, Mathai D, Bell JM, Jones RN, Mendes RE. Early dissemination of NDM-1- and OXA-181-producing Enterobacteriaceae in Indian hospitals: report from the SENTRY Antimicrobial Surveillance Program, 2006-2007. Antimicrob Agents Chemother 2011;55:1274ŌĆō1278PMID : 21189345.



82. Lee HK, Park YJ, Kim JY, et al. Prevalence of decreased susceptibility to carbapenems among Serratia marcescens, Enterobacter cloacae, and Citrobacter freundii and investigation of carbapenemases. Diagn Microbiol Infect Dis 2005;52:331ŌĆō336PMID : 15994052.


83. Rhee JY, Park YK, Shin JY, et al. KPC-producing extreme drug-resistant Klebsiella pneumoniae isolate from a patient with diabetes mellitus and chronic renal failure on hemodialysis in South Korea. Antimicrob Agents Chemother 2010;54:2278ŌĆō2279PMID : 20211897.



84. Kim MN, Yong D, An D, et al. Nosocomial clustering of NDM-1-producing Klebsiella pneumoniae sequence type 340 strains in four patients at a South Korean tertiary care hospital. J Clin Microbiol 2012;50:1433ŌĆō1436PMID : 22259206.



85. Roh KH, Lee CK, Sohn JW, Song W, Yong D, Lee K. Isolation of a Klebsiella pneumoniae isolate of sequence type 258 producing KPC-2 carbapenemase in Korea. Korean J Lab Med 2011;31:298ŌĆō301PMID : 22016687.



86. European Antimicrobial Resistance Surveillance Network (EARS-Net) [Internet]. European Centre for Disease Prevention and Control. c2012;cited 2012 Mar 20. Solna, Sweden: European Centre for Disease Prevention and Control, Available from: http://www.ecdc.europa.eu/en/activities/surveillance/ears-net.
87. Bertrand X, Dowzicky MJ. Antimicrobial susceptibility among gram-negative isolates collected from intensive care units in North America, Europe, the Asia-Pacific Rim, Latin America, the Middle East, and Africa between 2004 and 2009 as part of the Tigecycline Evaluation and Surveillance Trial. Clin Ther 2012;34:124ŌĆō137PMID : 22154196.


88. Livermore DM. Multiple mechanisms of antimicrobial resistance in Pseudomonas aeruginosa: our worst nightmare? Clin Infect Dis 2002;34:634ŌĆō640PMID : 11823954.


89. Breidenstein EB, de la Fuente-Nunez C, Hancock RE. Pseudomonas aeruginosa: all roads lead to resistance. Trends Microbiol 2011;19:419ŌĆō426PMID : 21664819.


90. Kolayli F, Gacar G, Karadenizli A, Sanic A, Vahaboglu H. Study Group. PER-1 is still widespread in Turkish hospitals among Pseudomonas aeruginosa and Acinetobacter spp. FEMS Microbiol Lett 2005;249:241ŌĆō245PMID : 16006075.


91. Naas T, Poirel L, Karim A, Nordmann P. Molecular characterization of In50, a class 1 integron encoding the gene for the extended-spectrum beta-lactamase VEB-1 in Pseudomonas aeruginosa. FEMS Microbiol Lett 1999;176:411ŌĆō419PMID : 10427724.


92. Cornaglia G, Giamarellou H, Rossolini GM. Metallo-╬▓-lactamases: a last frontier for ╬▓-lactams? Lancet Infect Dis 2011;11:381ŌĆō393PMID : 21530894.


93. Crespo MP, Woodford N, Sinclair A, et al. Outbreak of carbapenem-resistant Pseudomonas aeruginosa producing VIM-8, a novel metallo-beta-lactamase, in a tertiary care center in Cali, Colombia. J Clin Microbiol 2004;42:5094ŌĆō5101PMID : 15528701.



94. Silva FM, Carmo MS, Silbert S, Gales AC. SPM-1-producing Pseudomonas aeruginosa: analysis of the ancestor relationship using multilocus sequence typing, pulsed-field gel electrophoresis, and automated ribotyping. Microb Drug Resist 2011;17:215ŌĆō220PMID : 21332364.


95. Pitt TL, Livermore DM, Pitcher D, Vatopoulos AC, Legakis NJ. Multiresistant serotype O 12 Pseudomonas aeruginosa: evidence for a common strain in Europe. Epidemiol Infect 1989;103:565ŌĆō576PMID : 2514112.



96. Lee K, Park AJ, Kim MY, et al. Metallo-beta-lactamase-producing Pseudomonas spp. in Korea: high prevalence of isolates with VIM-2 type and emergence of isolates with IMP-1 type. Yonsei Med J 2009;50:335ŌĆō339PMID : 19568593.



97. Seok Y, Bae IK, Jeong SH, Kim SH, Lee H, Lee K. Dissemination of IMP-6 metallo-╬▓-lactamase-producing Pseudomonas aeruginosa sequence type 235 in Korea. J Antimicrob Chemother 2011;66:2791ŌĆō2796PMID : 21933788.


98. Gurung M, Moon DC, Tamang MD, et al. Emergence of 16S rRNA methylase gene armA and cocarriage of bla(IMP-1) in Pseudomonas aeruginosa isolates from South Korea. Diagn Microbiol Infect Dis 2010;68:468ŌĆō470PMID : 20926221.


99. Poirel L, Nordmann P. Carbapenem resistance in Acinetobacter baumannii: mechanisms and epidemiology. Clin Microbiol Infect 2006;12:826ŌĆō836PMID : 16882287.


100. Turton JF, Ward ME, Woodford N, et al. The role of ISAba1 in expression of OXA carbapenemase genes in Acinetobacter baumannii. FEMS Microbiol Lett 2006;258:72ŌĆō77PMID : 16630258.


101. Poirel L, Figueiredo S, Cattoir V, Carattoli A, Nordmann P. Acinetobacter radioresistens as a silent source of carbapenem resistance for Acinetobacter spp. Antimicrob Agents Chemother 2008;52:1252ŌĆō1256PMID : 18195058.



102. Park YK, Choi JY, Jung SI, et al. Two distinct clones of carbapenem-resistant Acinetobacter baumannii isolates from Korean hospitals. Diagn Microbiol Infect Dis 2009;64:389ŌĆō395PMID : 19631092.


103. Walsh TR. Clinically significant carbapenemases: an update. Curr Opin Infect Dis 2008;21:367ŌĆō371PMID : 18594288.


104. Lee K, Yum JH, Yong D, et al. Novel acquired metallo-beta-lactamase gene, bla(SIM-1), in a class 1 integron from Acinetobacter baumannii clinical isolates from Korea. Antimicrob Agents Chemother 2005;49:4485ŌĆō4491PMID : 16251286.



105. Fournier PE, Vallenet D, Barbe V, et al. Comparative genomics of multidrug resistance in Acinetobacter baumannii. PLoS Genet 2006;2:e7. PMID : 16415984.



106. Adams MD, Goglin K, Molyneaux N, et al. Comparative genome sequence analysis of multidrug-resistant Acinetobacter baumannii. J Bacteriol 2008;190:8053ŌĆō8064PMID : 18931120.



107. Beceiro A, Llobet E, Aranda J, et al. Phosphoethanolamine modification of lipid A in colistin-resistant variants of Acinetobacter baumannii mediated by the pmrAB two-component regulatory system. Antimicrob Agents Chemother 2011;55:3370ŌĆō3379PMID : 21576434.



108. Henry R, Vithanage N, Harrison P, et al. Colistin-resistant, lipopolysaccharide-deficient Acinetobacter baumannii responds to lipopolysaccharide loss through increased expression of genes involved in the synthesis and transport of lipoproteins, phospholipids, and poly-╬▓-1,6-N-acetylglucosamine. Antimicrob Agents Chemother 2012;56:59ŌĆō69PMID : 22024825.



109. Hornsey M, Ellington MJ, Doumith M, et al. AdeABC-mediated efflux and tigecycline MICs for epidemic clones of Acinetobacter baumannii. J Antimicrob Chemother 2010;65:1589ŌĆō1593PMID : 20554571.


110. Chisholm SA, Mouton JW, Lewis DA, Nichols T, Ison CA, Livermore DM. Cephalosporin MIC creep among gonococci: time for a pharmacodynamic rethink? J Antimicrob Chemother 2010;65:2141ŌĆō2148PMID : 20693173.


111. Lee SG, Lee H, Jeong SH, et al. Various penA mutations together with mtrR, porB and ponA mutations in Neisseria gonorrhoeae isolates with reduced susceptibility to cefixime or ceftriaxone. J Antimicrob Chemother 2010;65:669ŌĆō675PMID : 20093260.



112. Chisholm SA, Alexander S, Desouza-Thomas L, et al. Emergence of a Neisseria gonorrhoeae clone showing decreased susceptibility to cefixime in England and Wales. J Antimicrob Chemother 2011;66:2509ŌĆō2512PMID : 21846672.


113. Bignell C, Fitzgerald M. Guideline Development Group. British Association for Sexual Health and HIV UK. UK national guideline for the management of gonorrhoea in adults, 2011. Int J STD AIDS 2011;22:541ŌĆō547PMID : 21998172.


114. Ohnishi M, Golparian D, Shimuta K, et al. Is Neisseria gonorrhoeae initiating a future era of untreatable gonorrhea? Detailed characterization of the first strain with high-level resistance to ceftriaxone. Antimicrob Agents Chemother 2011;55:3538ŌĆō3545PMID : 21576437.



115. Unemo M, Golparian D, Nicholas R, Ohnishi M, Gallay A, Sednaoui P. High-level cefixime- and ceftriaxone-resistant Neisseria gonorrhoeae in France: novel penA mosaic allele in a successful international clone causes treatment failure. Antimicrob Agents Chemother 2012;56:1273ŌĆō1280PMID : 22155830.



116. Yahav D, Lador A, Paul M, Leibovici L. Efficacy and safety of tigecycline: a systematic review and meta-analysis. J Antimicrob Chemother 2011;66:1963ŌĆō1971PMID : 21685488.


117. Livermore DM. Doripenem: antimicrobial profile and clinical potential. Diagn Microbiol Infect Dis 2009;63:455ŌĆō458PMID : 19302929.


118. Livermore DM, Mushtaq S, Ge Y, Warner M. Activity of cephalosporin CXA-101 (FR264205) against Pseudomonas aeruginosa and Burkholderia cepacia group strains and isolates. Int J Antimicrob Agents 2009;34:402ŌĆō406PMID : 19428220.


119. Sader HS, Rhomberg PR, Farrell DJ, Jones RN. Antimicrobial activity of CXA-101, a novel cephalosporin tested in combination with tazobactam against Enterobacteriaceae, Pseudomonas aeruginosa, and Bacteroides fragilis strains having various resistance phenotypes. Antimicrob Agents Chemother 2011;55:2390ŌĆō2394PMID : 21321149.



120. Livermore DM, Mushtaq S, Warner M, et al. Activities of NXL104 combinations with ceftazidime and aztreonam against carbapenemase: producing Enterobacteriaceae. Antimicrob Agents Chemother 2011;55:390ŌĆō394PMID : 21041502.



121. Armstrong ES, Miller GH. Combating evolution with intelligent design: the neoglycoside ACHN-490. Curr Opin Microbiol 2010;13:565ŌĆō573PMID : 20932796.


122. Livermore DM, Mushtaq S, Ge Y. Chequerboard titration of cephalosporin CXA-101 (FR264205) and tazobactam versus beta-lactamase-producing Enterobacteriaceae. J Antimicrob Chemother 2010;65:1972ŌĆō1974PMID : 20595207.


123. Baker SJ, Tomsho JW, Benkovic SJ. Boron-containing inhibitors of synthetases. Chem Soc Rev 2011;40:4279ŌĆō4285PMID : 21298158.


124. Xiao XY, Hunt DK, Zhou J, et al. Fluorocyclines. 1.7-fluoro-9-pyrrolidinoacetamido-6-demethyl-6-deoxytetracycline: a potent, broad spectrum antibacterial agent. J Med Chem 2012;55:597ŌĆō605PMID : 22148514.


125. Masterton RG. The new treatment paradigm and the role of carbapenems. Int J Antimicrob Agents 2009;33:105ŌĆō110PMID : 18848436.


126. Kumar A. Optimizing antimicrobial therapy in sepsis and septic shock. Crit Care Clin 2009;25:733ŌĆō751PMID : 19892250.


127. Soo Hoo GW, Wen YE, Nguyen TV, Goetz MB. Impact of clinical guidelines in the management of severe hospital-acquired pneumonia. Chest 2005;128:2778ŌĆō2787PMID : 16236955.


128. Gaibani P, Rossini G, Ambretti S, et al. Blood culture systems: rapid detection-how and why? Int J Antimicrob Agents 2009;34(Suppl 4):S13ŌĆōS15PMID : 19931809.


Figure┬Ā1
Outer layers of the Gram-negative cell. To reach its target, the antibiotic must diffuse across the outer membrane, normally via porins, which form water-filled channels and exclude large or hydrophobic molecules. The drug must then evade periplasmic ╬▓-lactamases and efflux pumps; depending on its target, it may also need to cross the cytoplasmic membrane, often by energy-dependent uptake.
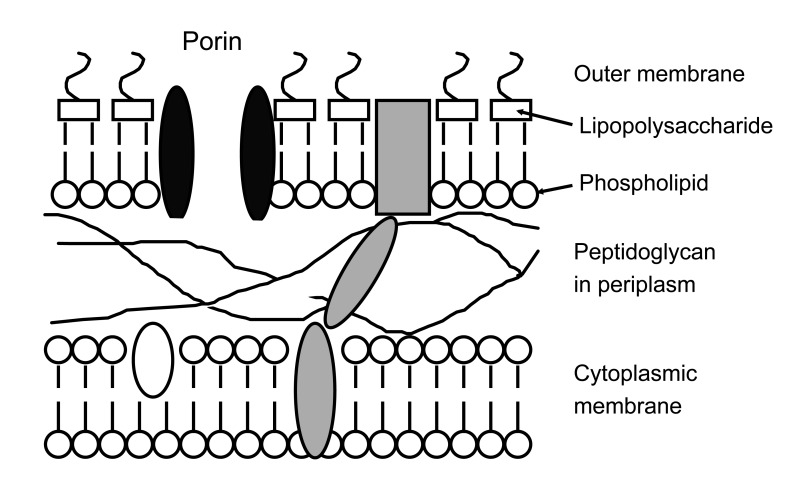
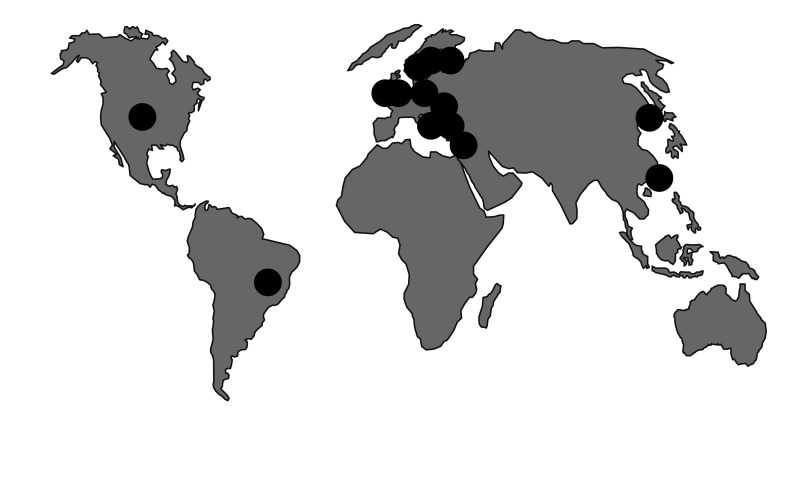
Figure┬Ā2
International spread of Escherichia coli ST131 [25,40] . This strain has played a major role in the dissemination of CTX-M-15 extended-spectrum ╬▓-lactamase (ESBL), but occasionally hosts other types. It occurs in Korea, where CTX-M-14 ESBL (not linked to a particular clone) is also widespread [48].
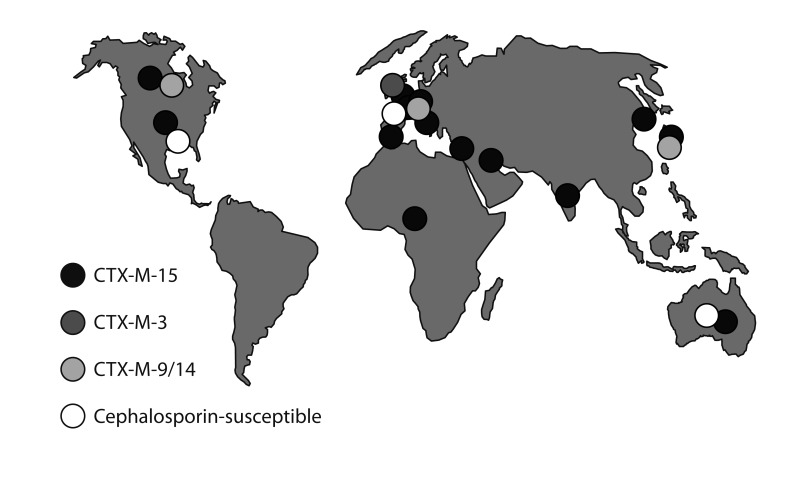
Figure┬Ā4
Spread of OXA-48 and related carbapenemases. Until 2007-2008, the OXA-48 enzyme was confined to Turkey. Carried by 50- and 70-kb plasmids, it has since spread to North Africa, the Middle East, and, increasingly Europe. A related enzyme, OXA-181, is circulating in India, but is rarer than the NDM-1 metallo-carbapenemase. These enzymes have not yet reached Korea.
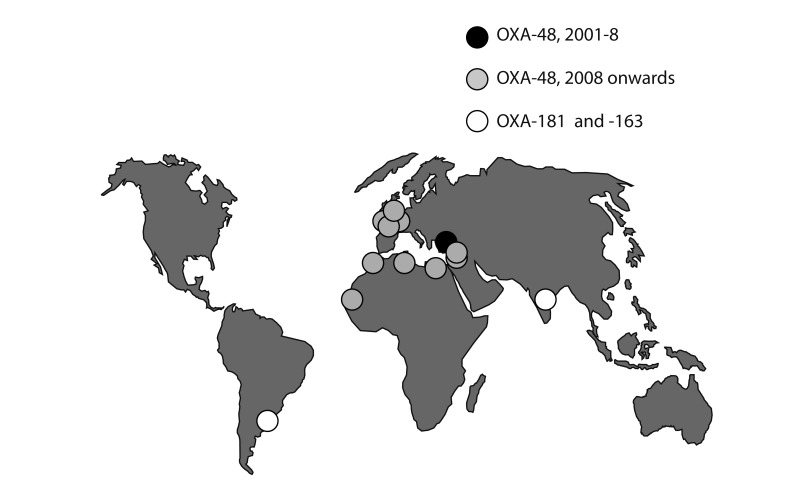
Figure┬Ā5
Reported outbreaks due to metallo-╬▓-lactamase-producing Pseudomonas aeruginosa. Most carbapenem resistance in P. aeruginosa, including in Korea, is due to porin loss, but carbapenemase producers occur and may be strongly persistent in some hospitals. Korea has a particular concern regarding a nationally distributed strain that harbors the IMP-6 enzyme [97].
Table┬Ā1
Consequences of selection of AmpC-derepressed mutants during cephalosporin treatment of Enterobacter infections

Cases exhibited selection of AmpC-derepressed mutants during cephalosporin treatment of Enterobacter infections; controls are those in whom cephalosporins were used successfully, without selection of resistance [16].
Table┬Ā2
Ability of ╬▓-lactamases to confer resistance
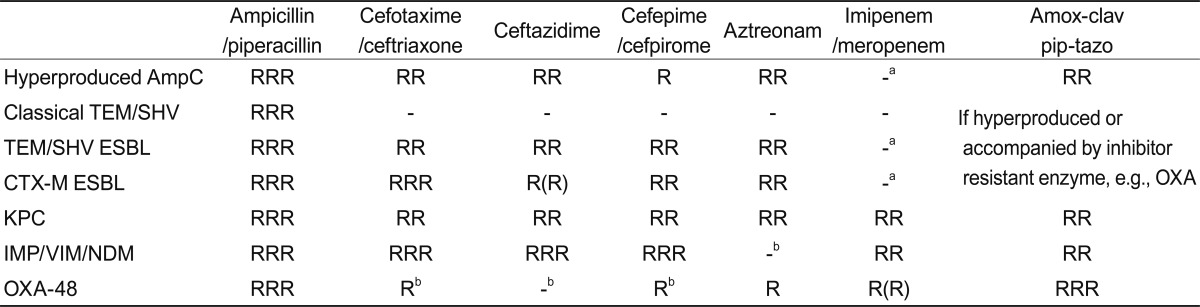
RRR, high-level resistance always seen; RR, resistance usually clear; R, MICs raised but may or may not exceed breakpoint; -, resistance not normally seen.
ESBL, extended-spectrum ╬▓-lactamase.
aResistance arises if combined with impermeability.
bResistance widespread as producers often also have ESBLs.
-
METRICS

- Related articles
-
Epidemiology and treatment of antimicrobialresistant gram-negative bacteria in Korea2018 March;33(2)



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print


