 |
 |
| Korean J Intern Med > Volume 17(4); 2002 > Article |
|
Abstract
Pancreatic endometrial cyst is an extremely rare instance of ectopic endometriosis that was first described by Marchevsky in 19841). A 21-yr-old woman with a history of epigastric pain and weight loss was found to have a cystic lesion in the pancreas on CT-scan. Under the tentative diagnosis of a pancreatic cystic neoplasm, partial pancreatectomy was performed. Histopathological examination of the specimen revealed cystic endometriosis. The clinicopathological features of the lesion are discussed and literature concerning this extremely rare lesion is reviewed.
Cystic lesions of the pancreas occasionally present problems in diagnosis and management, especially when found incidentally. Cystic lesions can be broadly classified as follows: congenital true cyst; primary cystic neoplasms; acquired cysts, including parasitic cysts and postinflammatory cysts (pseudocyst, retention cyst); extra-pancreatic cystic disorders masquerading as pancreatic cyst; and other miscellaneaus cystic lesion2). Endometriosis is defined as the presence of endometrial glands and stroma occurring outside of the uterine cavity3). It has been encountered in multiple sites such as the ovaries, fallopian tubes, uterus, peritonium, uterine ligaments, rectovaginal septum, cervix, vagina, vulva, skin, umbilicus, incisional scars, hernia scars, bladder wall, kidney, pelvic lymph nodes, extremities, lung, pleura, large intestine, rectum, appendix, gallbladder, stomach, and spleen3–5). To our knowledge, endometriosis of the pancreas is an extremely rare disease. We report a patient with a cyst in the body of the pancreas that on pathologic examination proved to be lined by the ectopic endometrial tissue.
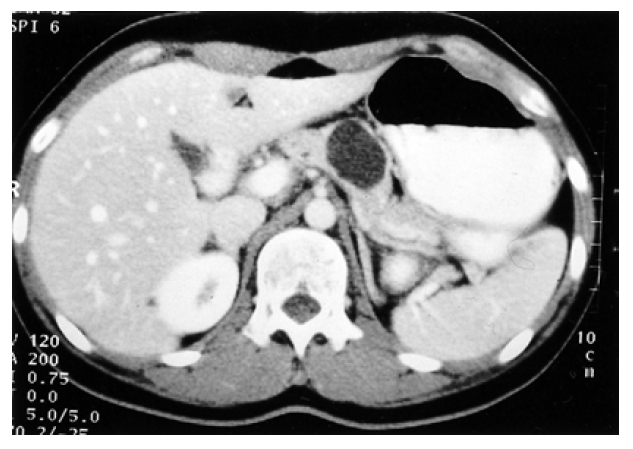
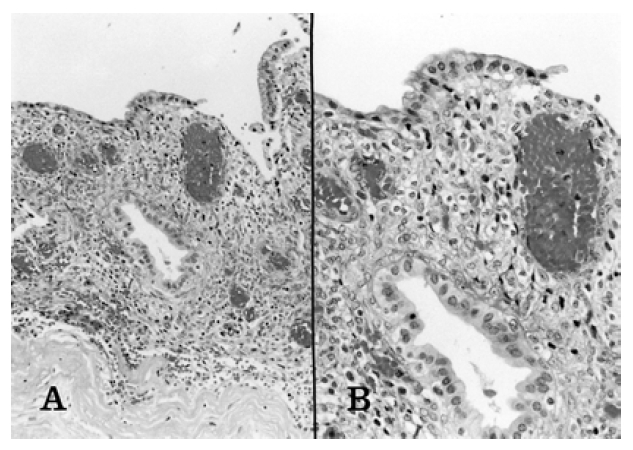
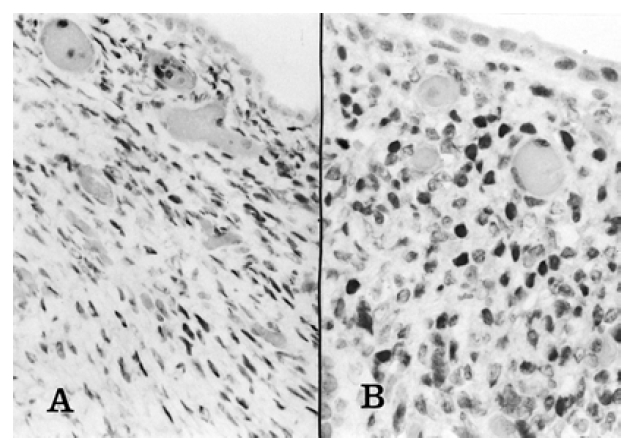
A 21-yr-old Korean woman was admitted to the hospital with a recurrent epigastric pain and 8 kg weight loss over a one year period. She denied a history of hepato-biliary disease, alcoholism, previous surgery, traumatic abdominal injury or cyclic abdominal pain. The familial history was unremarkable. Her menstrual history was normal. On admission, her blood pressure was 120/80 mmHg, pulse 80/min, temperature 36.5°C and respirations 20/min. On examination, there was no abnormally palpable mass in her abdomen. Laboratory data on admission was as follows : hemoglobin 12.4 g/dL, hematocrit 37.3%, white blood cell count 5,500/mm3, platelet 300,000/mm3, total protein 6.4 g/dL, albumin 4.2 g/dL, total bilirubin 0.7 mg/dL, AST 10 IU/L, ALT 9 IU/L, alklaine phosphatase 135 IU/L, GGT 12 IU/L, amylase 60 U, CEA 1.24 ng/dL, CA-125 8.71 U/mL, CA 19-9 54.34 U/mL, AFP 0.4 ng/mL, CA 15-3 9.09 U/mL. Computed tomography of the abdomen showed a 3.5×4.0 cm-sized, oval-shaped low density cystic mass involving the body of the pancreas with bulging-out contour. This cystic mass showed distinct capsular enhancement and some suspicious internal spetations. There was no definite pancreatic duct dilatation (Figure 1). With a preoperative diagnosis of mucous cystic adenoma, the patient underwent partial pancreatectomy. There was a 4 cm cyst involving the body of the pancreas. The remainder of the pancreas appeared normal. Gross finding of surgical specimen revealed a 4.0×4.0×4.5 cm well-circumscribed cyst located within the pancreatic stroma. The cyst was lined by a smooth, trabeculated, grey-brown wall with focal areas of hemorrhage. On microscopy, the cyst was covered with fibrous tissue containing endometrial elongated glands consisting of cuboidal and columnar cells with basal nuclei and amphophilic cytoplasm. The glands were admixed with well-vascularized spindle cell stroma containing multiple areas of focal hemorrhage and hemosiderin deposition in macrophage (Figure 2). Immunohistochemical examinations showed positive tests to antibodies to the estrogen receptor-related protein (ER) and the nuclear progesteron receptor protein (PR) (Figure 3). The remainder of the pancreas was unremarkable. The postoperative course was uneventful.
Extrapelvic endometriosis refers to endometropic implants found in other areas of the body, including the vagina, vulva, cervix and perineum, in the inguinal canal, the urinary system, the gastrointestinal tracts, pulmonary structures, extremities, skin and central nervous system. The epidemiology, diagnosis and natural history of extrapelvic endometriosis appear to differ somewhat from endometriosis. It remains uncertain whether lesions distant from pelvic sites even represent the same disease process and whether the same diagnostic and treatment modalities are appropriate. The true prevalence of extrapelvic endometriosis is unknown6). In gastrointestinal tract endometriosis, the intestines are involved in 5–15% of all the cases of endometriosis, and the gastrointestinal tract is the most common site of extrapelvic disease7–9). Cystic endoteriosis of the pancreas is very rare and has been mentioned in the literature only four times. Other reports have shown endometrial cysts in the pancreatic tail1, 10–12). But, in our presented case, an endometrial cyst was located in the body of the pancreas. Endometriosis is defined as the presence of endometrial glands and stroma occurring outside of the uterine cavity3). Histopathological demonstration of endometrial tissue from various parts of the mass is desirable for confirmation of the diagnosis. In our presented case, correlation of the histopathological findings to the endometriosis could be demonstrated. Immunohistochemical investigations of a previous report with antibodies to the estrogen receptor-related-protein and the nuclear progeteron receptor protein was negative. In our case however, immunostaining were positive. The possible pathogenesis of endometriosis in multiple organs has been the subject of controversy and numerous theories. For more than 70 years, various theories have been promulgated to explain the pathogenesis of endometriosis. Interest in the genesis of the endometric lesion has been a focus since the earliest studies. More recently, investigator implants result from menstrual flow through the fallopian tube14). The theory of direct extension proposes that endometriosis results from direct invasion of the ectopic endometrium through the uterine musculature13). Spread of endometrial glands and stroma through muscle fibers or along lymphatic and venous channels may, in fact, be a progenitor of adenomyosis, but the relationship of direct endometrial spread to endometriotic implants is not well established15). The theory of coelomic metaplasia holds that endometriosis developed from metaplasia of cells lining the pelvic peritonium. This is based on embryologic studies demonstrating that mullerian ducts, germinal epithelium of the ovary and pelvic peritoneum are all derived from the same embryologic precusor15). An attractive component of the coelomic metaplasia theory is that it can account for the occurrence of endometriosis anywhere in the abdominal cavity13). The theory of lymphatic and vascular metastasis had esuggested that endometriosis could result from lymphatic and hematogenous dissemination of endometrial cells16). It has been offered as an explanation for rare cases of endometriosis occurring in locations remote from the peritoneal cavity. In addition to pleural tissue, endometriosis has been reported in pulmonary parenchyma, bone, biceps muscle, peripheral nerves and the brain17–19). The composite theory of the histogenesis of endometriosis combines the implantation, vascular/lymphatic metastasis and direct extension theories20). In a previous report, the pancreas was located in the retroperitoneum and had no clear connection with the peritoneal cavity, a finding that endometrial fragments could be transported through lymphatic or blood vessels into such distant sites1). Because there are protean manifestations of endometriosis, it is likely that several mechanisms are involved in the pathogenesis. Further clinical and experimental investigations are needed to evaluate the pathogenesis of endometriosis occurring in unusual sites.
REFERENCES
1. Marchenvsheky AM, Zimmerman MJ, Aufses AH, Weiss H. Endometrial cyst of pancreas. Gastroenterology 86:1589–15911984.


5. Tornquist B. Endometriosis in vagina, vulvar and perineal scars. Acta Obstet Gynecol Scand 29:485–4891949.

7. Zwas FR, Lyon DT. Endometriosis. An important condition in clinical gastroenterology. Dig Dis Sci 36:353–3641991.


8. Forgren H, Lindhangen J, Melander S, et al. Colorectal endometriosis. Acta Chir Sand 149:431–4351983.
9. Meyer W, Kelvin F, Jones R. Diagnosis and surgical treatment of colonic endometriosis. Arch Surg 1979;114:169–175.


10. Sumiyoshi Y, Yamashita Y, Maekawa T, Sakai T, Shirakusa T. A case of hemorrhagic cyst of the pancreas resembling the cystic endometriosis. Int Surg 85:67–702000.

11. Verbeke C, Harle M, Sturm J. Cyst endometriosis of the upper abdominal organs:report on three cases and review of the literature. Pathol Res Pract 192:300–3041996.


12. Goswami AK, Sharma SK, Tandon SP, Malik N, Mathur RP, Malik AK, Bapna BC. Pancreatic endometriosis presenting as a hypovascular renal mass. J Urol 135:112–1131986.


13. Craig AW. Current concepts in the pathogenesis of endometriosis. Clin Obstet Gynecol 1:42:566–5851999.
14. Sampson J. Peritoneal endometriosis due to the menstrual dissemination of endometrial tissue into the peritoneal cavity. Am J Obstet Gynecol 14:422–4691927.

15. Gruenwald P. Origin of endometriosis from the mesenchyme of the coelomic walls. Am J Obstet Gynecol 44:470–4741942.

17. Cassina PC, Hauser M, Kacl G, et al. Catamenial hemoptysis. Diagnosis with MRI. Chest 111:1447–14501997.


-
METRICS

- Related articles
-
A Case of Idiopathic Granulomatous Hypophysitis2012 September;27(3)
A case of scar sarcoidosis2008 December;23(4)
A Case of Cushing's Syndrome Presenting as Endometrial Hyperplasia2008 March;23(1)
A Case of Pulmonary Vein Tumor Presenting as a Left Atrial Mass2007 March;22(1)
A Case of Primary Pancreatic Non-Hodgkin's Lymphoma2006 June;21(2)



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print


